The Critical Role of Dopamine in the Evolution of Human Intelligence and Thermal Tolerance
 https://orcid.org/0000-0002-3159-6578
https://orcid.org/0000-0002-3159-6578
Department of Psychology, The University of Texas at San Antonio, One UTSA Circle, San Antonio TX, USA
Abstract. Modern humans are unique among anthropoids in many key features, including our advanced intelligence, large brain-body size, thermal tolerance, and endurance capability. The objective of this theoretical review is to update the theory of Previc (1999) postulating the importance of dopamine in human evolution by synthesizing newer findings concerning dopamine’s role in human intellectual and endurance capabilities. Recent evidence further supports the putative role of dopamine in advanced human intelligence (especially cognitive flexibility) and thermal tolerance and endurance. One key breakthrough is a collection of recent studies demonstrating a uniquely human dopaminergic innervation of the striatum and prefrontal cortex—both essential to human cognition. Another potentially important finding is the human-specific mutation of an enhancer to the EN1 gene that controls eccrine gland formation and plays a major role in the development of dopaminergic brain systems. A plausible evolutionary scenario is put forth in which the enhanced thermal capabilities linked to dopaminergic evolution may have gradually led to the enhanced intellects of modern humans.
Keywords: dopamine, intelligence, endurance, evolution, human
Introduction
Humans are unique among primates and even most mammals in several key respects, most importantly our advanced intelligence and thermal endurance capacity during exercise. When these various traits emerged during hominin evolution remains speculative, but the theory presented in this paper—updated from that of Previc (1999)—is that, because of their synergistic relationships, most traits must have emerged concurrently between~ 2.8 million years ago (mya) (just prior to the emergence of Homo habilis) and ~1.2 mya (after the emergence of Homo erectus).
One key synergy is in the area of human thermal tolerance, which largely depends on the combined advantages of reduced body hair cover and predominantly eccrine sweating; one without the other would be much less advantageous (Lieberman 2015). Other linkages such as bipedalism and increased stature would further enhance thermal tolerance by decreasing exposure to solar irradiance (Wheeler 1985), and thermal exchange via the eccrine glands would be aided by the increased surface-to-mass ratio (Allen’s rule) (Tilkens et al. 2007). In turn, the ability to travel long distances in the heat of the midday sun would allow early humans to scavenge and possibly chase-hunt for meat while predators would be resting to avoid hyperthermia (Carrier 1984; Bortz 1985; Lieberman 2015). The switch to a tyrosine-rich carnivorous diet would increase the brain-body ratio due to the combination of a larger brain (Previc 1999) and reduced intestinal mass characteristic of carnivores (Henneberg and Sarafis 1998) and lead to a concomitant increase in dopamine-mediated advanced intelligence.
This paper will postulate that all of these unique features of humans were caused, causal to, or correlated with increases in the neurotransmitter dopamine and its precursor tyrosine during human evolution. The key roles of dopamine in the evolution of advanced intelligence and thermal tolerance were previously postulated (Previc 1999), but more recent findings to be reviewed here provide a more complete and compelling synthesis of the dopaminergic role in these interleaved components of human evolution. For one, massive and unique increases in dopamine innervation and activity in the human striatum and frontal lobes of humans—key areas involved in higher-level cognition—have been recently found. The importance of dopamine in speech, abstract representations, creativity and a host of other key intellectual functions has also been further demonstrated (see next section). In terms of thermal tolerance, new findings have documented the role of dopamine in creating hair loss and the genetic overlap between the formation of both brain dopaminergic systems and eccrine sweat glands during human evolution (see “The Role of Dopamine in Human Thermal Endurance—New Findings”). And, the importance of meat consumption—previously documented by cut marks on and stone-flaked tools near animal bones in early Homo sites—has been supported by genetic analyses of human tapeworms (Hoberg et al. 2001) and pH levels (Beasley et al. 2015) in the human stomach and gut.
Brain Dopamine and Human Intelligence – New Findings Dopamine and Human Intelligence
Previc (1999) theorized the importance of neurochemical changes involving the neurotransmitter dopamine in the evolution of human intelligence, highlighting six key traits essential to human intelligence and human language—motor planning, working memory, cognitive flexibility, abstract reasoning, temporal analysis/sequencing, and generativity. Previc (1999) argued against the role of brain size and genetic factors in determining human intelligence, instead emphasizing the importance of epigenetic transmission during human evolution. He noted the role of prenatal factors in the creation of brain lateralization in humans (see also Previc 1991), which in most individuals is associated with profound differences in intellectual and other abilities between the left and right hemispheres despite their identical anatomical size and the absence of any known genetic influence. The importance of nongenetic factors in intelligence is supported by recent genome-wide analyses that have consistently shown that only about 5% of the variance in intelligence as measured through standard tests is directly attributable to genes (Benyamin et al. 2014; Davies et al. 2016; Sniekers et al. 2017; Kaminski et al. 2018), with many of the key ones involved in dopaminergic synaptic transmission (DeYoung et al. 2011; Zabelina et al. 2016; Kaminski et al. 2018). Indeed, epigenetic and genetic influences on just six genes involving the synthesis, transmission, breakdown, and re-uptake of dopamine at the synapse can predict much of the variance in childhood intelligence accounted by genome-wide models (Kaminski et al. 2018).
The critical role of dopamine in key “executive” abilities such as working memory, creativity and language has also been confirmed in numerous recent studies and reviews: working memory (D’Esposito and Postle 2015; Matzel and Sauce 2023); creativity/divergent thinking (Takeuchi et al. 2014; Lhommee et al. 2014; Zabelina et al. 2016); cognitive flexibility and set-shifting (Ko et al. 2008; Garcia-Garcia et al. 2010); speech (Simoyan et al. 2013; Fuertinger et al. 2018; Rusz et al. 2024); future goals and planning (Hart et al. 2024); and time perception and future orientation (Mitchell et al. 2018). An underlying component of dopamine’s role in most of these abilities is cognitive flexibility—the ability to update processing and actions on the basis of new information and contexts. For example, working memory requires constant updating and attentional disengagement when new information is processed (Matzel and Sauce 2023), divergent thinking requires switching to new mental models and approaches (Palmiero et al. 2022), and choice and switching of goals is correlated with cognitive flexibility scores (Leclercq et al. 2023). Cognitive flexibility stems from dopamine’s dual role in the prefrontal cortex, promoting the inhibition required to stop ongoing behavior and the excitation required to engage in new behavior and processing (Mitchell et al. 2018; Di Domenico and Mapelli 2023). This inhibition is carried out by interneurons in the prefrontal cortex, which are preferentially innervated by dopamine in humans (see next section).
Unique Dopaminergic Features of the Human Brain
As of 1999, there was little direct evidence that dopaminergic innervation of the human brain had selectively increased relative to other hominids. A lone study by Ikemoto et al. (1997) showed many more neurons staining for dopa-decarboxylase, the enzyme that converts the precursor L-dopa to dopamine, in the striatum of humans relative to monkeys. But the evolutionary significance of this finding was unclear since neither chimpanzee nor gorilla brains were used as comparisons.
Beginning in 2009, a series of studies carried out by Raghanti and colleagues (2009) found large increases in dopaminergic innervation in areas of the human brain critical to intelligence. All of these studies used stains for tyrosine hydroxylase (TH), the enzyme responsible for converting tyrosine to L-dopa, the immediate precursor to dopamine that crosses the blood-brain barrier.
Raghanti et al. (2009) showed substantial TH staining in human but not chimpanzee and gorilla prefrontal interneurons, suggesting selective catecholaminergic activity in humans. However, because TH is a precursor to noradrenaline via dopamine, no firm conclusion could be drawn as to the selectivity of dopaminergic innervation of human prefrontal cortex. Raghanti et al. (2016) later showed, using TH staining and a measure of axonal length to neuronal density, over twice as much dopaminergic innervation in the medial caudate portion of the dopamine-rich human dorsal striatum compared to that of gorillas and chimpanzees. The noradrenergic contribution to the TH increase in this case could be ruled out because noradrenaline concentrations in the human caudate are ~50 times less than those of dopamine (Tong et al. 2006), despite both catecholamines constituting only a tiny overall percentage of the total neurons in the caudate nucleus and dorsal striatum, the latter containing both the caudate and putamen. The reason for the huge difference in catecholamine concentrations is because of the extraordinary arborization of dopamine axons emanating from the midbrain substantia nigra, with a single nigrostriatal axon on average contacting up to 75,000 striatal neurons, or 2.7% of the total volume of the neostriatum of the rat (Matsuda et al. 2009). Tyrosine hydroxylase staining in other subcortical dopamine-rich regions was generally greater in humans but less dramatically so than in the medial caudate nucleus. The differences between humans and other hominids in dopamine innervation in the medial caudate were especially impressive in Raghanti et al. (2016) due to the remarkably low variability in human TH levels in that region (Raghanti et al. 2016, Fig. 3).
Sousa et al. (2017) reported elevated TH expression as well as that of dopamine decarboxylase in a wide range of brain areas in humans relative to chimpanzees. The largest differences overall were in the dorsal striatum, but there were also major differences in prefrontal interneurons, with almost no ape prefrontal interneurons staining for TH. In yet another study using TH staining, Hirter et al. (2021) showed significantly greater dopaminergic innervation in the ventral striatum (ventral palladium and nucleus accumbens) of humans relative to chimpanzees and gorillas. Finally, Ma et al. (2022) showed, based on the relative staining for TH vs. somatostatin (SST), that a remarkable substitution of dopamine for SST occurred during human evolution in prefrontal interneurons, with only 2.4% of human TH-stained interneurons co-localized with SST, in contrast to almost 50% in the macaque monkey. The percentage of TH staining could not even be calculated in the interneurons of chimpanzees and gorillas because it was essentially nonexistent (Ma et al. 2022), in line with the findings of Raghanti et al. (2009) and Sousa et al. (2017). The significance of this intriguing substitution of SST for dopamine in the very interneurons presumed to be involved in the key ability of cognitive flexibility (see earlier section) remains unclear, however.
Doll et al. (2024) in their Figure 1b summarize the expansion of dopaminergic systems in the human brain that has been documented using TH staining as well as staining for dopa decarboxylase and DAT, which reabsorbs synaptic dopamine back into presynaptic neurons. Additional evidence of a dopaminergic-intelligence link in humans are the significant correlations between volume of the dopamine-rich dorsal striatum and intelligence (Grazioplene et al. 2015; Kaminski et al. 2018). (By contrast, overall brain volume predicts only a small amount of the variance in human intelligence—Pietschnig et al. 2015.) These findings confirm the previously documented importance of the integrity of striatal-frontal circuits (Kling and Tucker 1967) and of the striatum in particular (Goldman and Rosvold 1972) for primate cognitive development. Moreover, selective destruction of dopamine neurons (Brozoski et al. 1979) or blockade of D1 and D2 post-synaptic dopamine receptors (Ranganath and Jacob 2016) essentially replicates the cognitive deficits following lesioning of the prefrontal cortex.
In contrast to the expansion of dopaminergic innervation in the prefrontal cortex and striatum, expression of other major neurotransmitters such as norepinephrine, GABA, glutamate and acetylcholine appears either unchanged or even reduced in humans (Sousa et al. 2017; Raghanti et al. 2018). For example, TH staining in prefrontal interneurons did not co-locate with staining for dopamine beta-hydroxylase (Sousa et al. 2017), which converts dopamine into norepinephrine, indicating that brain levels of norepinephrine are conserved in hominin evolution. While serotonergic innervation may have increased in certain areas of the human brain (Raghanti et al. 2009; Saniotis et al. 2021), there is little evidence that serotonin contributes to the advanced human cognitive skills listed by Previc (1999), as it even opposes dopamine action in the striatum and prefrontal cortex during learning and memory (Luciana et al. 1998; Daw et al. 2002; Olvera-Cortes et al. 2008). Also, unlike dopamine, serotonin in most humans does not more densely innervate the left hemisphere housing speech and analytical intelligence, nor can the hyperthermic action of serotonin (Tormoehlen and Rusyniak 2018) be reconciled with the greater thermal tolerance and endurance capabilities developed in early hominin evolution (see next section). However, brain serotonin does foster social cooperation and other prosocial behaviors (Crockett 2009), which may have facilitated the use of language and contributed to cultural and technological exchange during later human evolution (Raghanti et al. 2018; Saniotis et al. 2021).
Finally, the large increases in dopamine innervation of the human prefrontal cortex and striatum overall parallel the greater dopaminergic activity in the left hemisphere of most humans (Larisch et al. 1998; Previc 1999; Cho and Strafella 2009)—the citadel of advanced human thought with its dominance in speech, grammar, motor sequencing, abstract reasoning, analytical intelligence, and other unique characteristics of human cognition (Previc 1999; Simonyan et al. 2013; Fuertinger et al. 2018; Rusz et al. 2024). At least two of these cognitive abilities—speech and cognitive flexibility—have been shown to involve dopamine circuits specifically in the left hemisphere (Ko et al. 2008; Simonyan et al. 2013; Fuertinger et al. 2018).
The Role of Dopamine in Human Thermal Endurance—New Findings
It has long been theorized that the emergence of Homo coincided with an enhanced ability to endure strenuous activity over large distances and tolerate heat, useful in persistence hunting or scavenging (Carrier 1984; Bortz 1985; Wheeler 1985; Bramble and Lieberman 2004; Lieberman 2015). It is still debated whether early humans engaged in chase hunting (more likely to be associated with endurance running), active scavenging, or passive scavenging (Pobiner 2020), but all of these would have required great energy expenditures in the midday heat, when other predators would be inactive. Regardless of the specific means of procuring the meat, the ability of early Homo to tolerate heat would have been critical to overcoming these energetic demands.
Besides its direct role in intellectual functioning, a crucial role of dopamine in humans is to prevent hyperthermia during endurance activity. Dopamine levels are elevated during high-intensity and endurance activity (Bortz et al. 1981; Marques et al. 1984; Tyler et al. 2023), and dopamine cells in tissue slices from the anterior hypothalamus, important in temperature regulation, are mostly sensitive to temperature increases (Scott and Boulant 1984). The importance of dopamine in preventing hyperthermia during endurance exercise has been documented in many past studies (e.g., Cox and Lee 1980; Marques et al. 1984; Lee et al. 1985; Bauer et al. 1989), and its benefits in sustaining endurance performance have been confirmed by more recent ones (Balthazar et al. 2010; Zheng and Hasegawa 2016). Recent clinical findings have also substantiated the important effects of reduced dopaminergic activity in hyperthermic syndromes such as the Parkinsonism Hyperpyrexia Syndrome (Newman et al. 2009; Linares et al. 2016; Coon and Low 2018) and the Neuroleptic Malignant Syndrome associated with anti-psychotic treatments in schizophrenia (Tormoehlen and Rusyniak 2018; Hirschbeck et al. 2022), both in contrast to the hypothermic effect of disinhibited dopamine activity in Huntington Disease (Weydt et al. 2018).
How dopamine transiently achieves its hypothermic action during endurance activity is unclear, since dopamine evidently does not directly control sweating (Shibasaki and Crandall 2010); rather, hyperhidrosis (excessive sweating) is part of the Dopamine Withdrawal Syndrome (Chaudhuri et al. 2015). Dopamine-induced vasodilation (Brodde 1982; Brown et al. 2007) is one potential mode of action, since shunting of the blood to extremities allows for more effective heat transfer (Charkoudian 2003). However, dopaminergic effects are complex in that they may be temperature dependent—hypothermic at high temperatures, hyperthermic at low ones (Brown et al. 2007)—and it has even been argued that there are beneficial effects of brain dopamine on endurance activity aside from its effects on temperatures (e.g., Balthazar et al. 2010; Zheng and Hasegawa 2016, Fig. 2). Previc (1999) speculated that dopamine’s role in motor behavior may activate a caudate-hypothalamic axis (Lee et al. 1985) during exercise that enhances oxygen metabolism and delivery in advance of an actual rise in temperature.
Previc (1999) proposed that the enhanced endurance capability of humans gave rise to a surge in brain dopamine during early human evolution, mainly due to the increase in meat consumption, which would have provided a substantially greater amount of tyrosine in the diet. The evidence for increased meat consumption beginning around 2 mya is compelling (Ferraro et al. 2013; Zink and Lieberman 2016; Pobiner 2020), although sampling biases may distort some of the archaeological findings (Barr et al. 2022). The evidence for meat-eating includes: 1) stone artifacts and bone cut marks in close proximity to early human remains, suggesting meat extraction from carcasses (Pobiner 2020); 2) the reduced size of the modern human large intestine (Henneberg and Sarafis 1998) and an increase in stomach acidity (Beasley et al. 2015), both associated with carnivorous diets; 3) changes in dentition (smaller teeth, especially canines; more bite strength in molars) that could have reflected the consumption of softer, processed meat (Dean and Cole 2013; Zink and Lieberman 2016); and 4) the evolution of human-specific tapeworms linked to African fauna around 1.7 mya (Hoberg et al. 2001). The increased tyrosine in the diet of early Homo would have major consequences in that it could have stimulated growth hormone (Tam et al. 2020), thereby increasing stature and brain volume (Yuan et al. 2020), which are significantly correlated (Taki et al. 2012). The effect of meat consumption on height has been demonstrated in past as well as recent large-scale studies (Suzuki 1981; Desmond et al. 2021; Mosites et al. 2017). The enhancement of growth hormone by tyrosine may be mediated by dopamine, although not all evidence is supportive. But it is clear that dopamine itself, in the brain and elsewhere, is dependent on tyrosine availability (Montgomery et al. 2003), as is cognitive capability, especially cognitive inhibition (Bloemendaal et al. 2018; Hase et al. 2015; Kuhn et al. 2019).
Despite dopamine’s important role in thermal tolerance, a key issue was left unanswered in Previc (1999): How could brain dopamine have been a major force in the evolution of human endurance if it was dependent on that endurance capability and the consequent increase in meat consumption? A possible solution to this conundrum is the recent finding that the ENGRAILED gene (EN1), a transcription factor that facilitates the formation of eccrine sweat glands (Aldea et al. 2021; Aldea and Kamberov 2022), underwent an evolutionary change in humans due to repeated human-specific mutations of a key enhancer, ECE18. These mutations likely contributed to increasing eccrine density in humans ~10-fold relative to chimpanzees (Aldea and Kamberov 2022), which facilitates the phenomenal ability of humans to eliminate up to one liter of water per hour (Smith et al. 2021) and 12 liters per day by means of evaporative cooling in the skin. It is generally acknowledged that 90% of heat loss occurs through the skin, mostly by radiation and convection; however, evaporative cooling through the eccrine glands becomes more critical at high temperatures (Wang et al. 2016). What is intriguing about the EN1 evolutionary progression is that this gene is also important in the development of midbrain dopaminergic systems (Simon et al. 2004; LePen et al. 2008; Nordstroma et al. 2018; Nouri and Awatramani 2017), the primary source of dopamine in the forebrain. EN1 is part of a constellation of genes and pathways with many diverse effects in the human body but which all interact to influence both sweating and exercise tolerance as well as the formation and sustenance of brain dopamine systems. These include NURR1 (Amoasii et al. 2019), the WNT family (Alves dos Santos and Smidt 2011), and especially FOXA1 (Ferri et al. 2007; Cui et al. 2012), the latter of which helps regulate both EN1 and NURR1 (Ferri et al. 2007). However, only EN1 via mutations of its enhancer ECE18 is confirmed to have undergone a major functional alteration in humans (Aldea et al. 2021).
Given the fact that each of these genes has multiple regulatory functions in human development, it is possible that the links between these key dopaminergic genes and sweating are coincidental. Or, the EN1 mutation may have affected dopamine and eccrine sweating independently, since eccrine function is largely controlled by cholinergic rather than dopaminergic transmission (Shibasaki and Crandall 2010). Regardless, mutations of an enhancer to a major gene regulating both sweat gland formation and dopaminergic brain development offers a tantalizing clue as to how dopamine and thermal tolerance linked up during human evolution.
In contrast, the reduced body hair cover may have been more directly influenced by dopamine in that dopamine agonists are known to decrease hair cover by stimulating catagen (Langan et al. 2013), which prevents the transformation of fine vellus hair into fully mature terminal hair. The role of dopamine in hair loss is demonstrated by the hair loss in >50% of all Parkinson’s patients receiving dopaminergic therapy (Lee et al. 2024). Decreased hair cover would combine with increased dopaminergic vasodilation (Brodde 1982; Brown et al. 2007) to dissipate heat through the skin via the eccrine glands. A final thermal advantage possibly conferred by the tyrosine-dopamine link is the elongation of the human body, which would decrease solar irradiance and increase surface-to-mass ratio, thereby enhancing dermal heat transfer. While Wheeler’s original theory posited that bipedalism per se—which preceded the evolution of the Homo genus—increased thermal tolerance and endurance, this remains a contentious issue (see David-Barrett and Dunbar (2016) vs. Ruxton and Wilkinson (2011)). But bipedalism associated with an elongated body would certainly have provided a greater means of dissipating heat, in line with Allen’s rule. Allen’s rule is supported by both experimental studies of limb length and metabolism (Tilkens et al. 2007) as well as the negative association between body height and risk of heat stroke (Taylor et al. 2024).
Many other changes in the human physique, discussed by Bramble and Lieberman (2004), may have simply been associated with the elongation of the body in humans. Others such as changes in dentition and the shape of the foot (Bramble and Lieberman 2004; Zink and Lieberman 2016) have less clear evolutionary origins, but epigenetic factors may have played a role in these as well (Quinn 2012; Khalaf et al. 2022). One adaptation that is more likely to have coincided with the loss of terminal body hair is the increased melanin content of the skin, which would have offset the dangers of increased solar irradiation due to the loss of hair cover (Jablonski 2021). Without such pigmentation, folate metabolism would be seriously impaired, threatening hominin reproductive success (Jablonski 2021; Lucock 2023). Melanin synthesis is influenced by many factors, but it is ultimately dependent on tyrosine and its conversion to L-dopa and then dopaquinone (Maranduca et al. 2019), consistent with evidence that L-dopa treatment for Parkinson’s disease has stimulating effects on melanogenesis (Bougea et al. 2019). In line with the hypothesized link between skin pigmentation and reduced hair cover during early Homo evolution, significant inverse correlations exist between skin pigmentation and hair follicle density and hair length in extant humans, with the former accounting for over 28% of the variance in follicle density (Dhugga et al. 2014).
Implications for Human Evolution
The linkage between EN1 and associated genes and dopaminergic brain development suggests that the mutations of the EN1-enhancer ECE18 may have been a catalyst for ensuing evolutionary changes in early Homo. Several rationales support the view that the enormous increase in eccrine sweat glands occurred before many other evolutionary changes such as decreased hair cover and increased stature. Of the three factors unique to human thermal tolerance, elongation of the body occurred later in the period from 2.5 to 1.5 mya, based on the skeletal record (Will et al. 2017). The other factor—reduced hair cover due to a retraction of terminal hair—may have occurred secondarily as well, for in the absence of copious sweating capabilities a loss of hair cover would have exposed humans to damaging solar radiation, with mixed thermoregulatory effects (David-Barrett and Dunbar 2016). (By contrast, the benefits of an increase in eccrine sweat glands would also have been limited without a concomitant decrease in hair cover (Lieberman 2015), but at least expanded eccrine sweating by itself would not have proven harmful). An additional reason for believing in the primacy of eccrine gland evolution is that greater eccrine activation and perhaps density is associated with hotter climates (Best and Kamilar 2018; Best et al. 2019), and the climate of the savanna was hotter at the beginning of hominin evolution than after 2.5 mya, when worldwide climate underwent a cooling trend (Stanley 1995). On the other hand, as the climate cooled and dried and forests in East Africa gave way to grassland, arboreal creatures such as the Australopithecines would have had more trouble adapting to the new climate than early Homo, the latter having a greater surface-to-mass area, reduced hair cover, and a more advantageous foot structure (Bramble and Lieberman 2004). Because EN1 influences both eccrine and hair follicle development, it cannot be ruled out that the eccrine increase and loss of hair cover occurred simultaneously; however, hair follicle density per se has not changed in humans relative to chimpanzees (Kamberov et al. 2018), and there is no evidence that EN1 promotes the catagen-induced loss of terminal hair.
One plausible evolutionary scenario is portrayed in Figure 1. An initial genetic adaptation involving ECE18 and the EN1 gene could have independently increased eccrine sweat gland density and dopaminergic brain innervation. The latter’s elevation would then contribute to reduced human hair cover and increased peripheral vasodilation to further enhance human thermal tolerance.
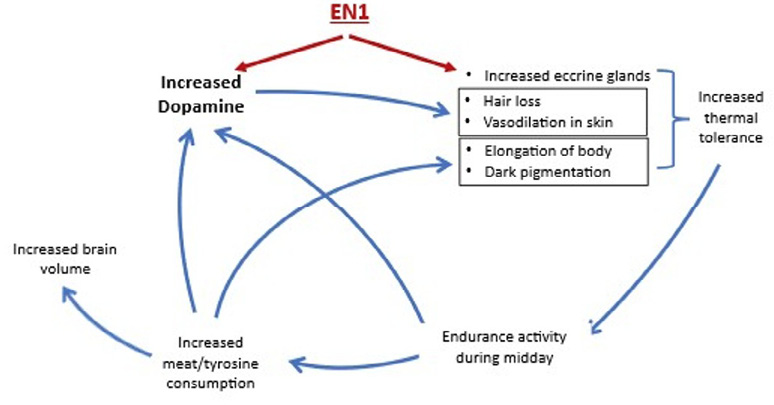
This facilitated the greater exploitation of the savanna grasslands by permitting travel over longer distances under thermally stressful conditions to procure meat, rich in tyrosine. The increase in tyrosine would have further increased brain dopamine, physical stature, and brain size along with increasing the melanin content of the human skin to protect it against the increased solar absorption due to the loss of hair cover. Once begun, the entire cyclical process could have been a gradual one, as suggested by Jablonski (2021, Fig. 1). A gradualist scenario is consistent with evidence that dopamine levels can be altered by epigenetic mechanisms and easily traverse the placenta, steadily accumulating over generations (see Previc 2009). The gradualist view of dopaminergic expansion is also consistent with recent re-appraisals of the cognitive capabilities of Homo erectus and archaic humans, given evidence of advanced lithic technology, abstract designs, food processing, and the use of fire dating back hundreds of thousands of years and, in some cases, even longer (Bednarik 1995; DeLouize et al. 2017; Clark and Henneberg 2021).
The putative rise in brain dopamine in conjunction with eccrine gland expansion and a less hairy and darkened skin is necessarily speculative and cannot yet be linked to a specific timeframe, since all scientists can currently decipher about soft tissue changes during human evolution is the difference between extant humans and chimpanzees. That divergence is believed to have occurred about 6–7 mya, and each species has evolved since then. The presence of the ECE18 mutations in the Neanderthal genome might push its existence as far back as back as 800 kya and perhaps somewhat plausibly even to the emergence of Homo erectus, but in the progression from Homo habilis to Homo erectus all that can be determined from the fossil and archaeological record is that more advanced tools were used to extract meat from carcasses, elongation of the body occurred, cranial capacity increased, and dentition evolved with ingestion of softer foods (Dean and Cole 2013). Aside from some limited genetic evidence related to tapeworm mutations, suggesting meat-eating as early as ~2 mya (Hoberg et al. 2001), there is little else available to capture precisely the evolutionary past other than theoretical analyses. Chemical composition in bone—especially relative zinc isotopes (Z66 to Z64) and carbon and nitrogen amino acid isotopes—offers promise in determining when greater carnivory occurred (Jaouen 2016; Jaouen et al. 2022; Larsen 2022), but these measures are difficult to interpret with remains dating back millions of years. So, definitive findings concerning when the putative rise in dopamine first occurred during hominin evolution is still lacking.
According to Previc (2009), the hominin progression leading from Homo habilis to Homo erectus was a major step in establishing the “dopaminergic mind”, but it was not the final stage. Despite large migrations of Homo erectus to different latitudes and regions with vastly different climates along with the thermal effect of clothing wear beginning around 170 kya (Toups et al. 2011), human intelligence, brain size, meat-eating, body height, sweat gland density, and many other distinguishing features of modern humans never retreated to pre-erectus levels. It may be presumed, as Previc (2009) argued, that dopamine levels in the human lineage continued to increase to the present, possibly epigenetically, as dopamine levels in individuals can be altered through diet, exercise, and psychological factors (e.g., stress) and can be easily passed on to offspring through placental transmission (Horackova et al. 2022). In the end, Homo sapiens acquired a large dopaminergic brain innervation and an impressive intelligence and thermal tolerance without parallel among mammals.
Conclusion
Recent findings have confirmed three of the major tenets of Previc (1999): the key roles of dopamine in human intelligence; the expansion of dopaminergic innervation of brain areas essential to human cognitive abilities; and the criticality of dopamine in thermal tolerance. Which uniquely human traits evolved after the divergence with chimpanzees is much better understood than how and when those traits evolved. But recent findings also offer a clearer glimpse than ever before as to what might plausibly constitute the progression of events during hominin evolution from Homo habilis to Homo erectus. Almost certainly, the neurotransmitter dopamine exerts a paramount influence on that evolutionary course.
References
Aldea D, Atsuta Y, Kokalari B., Schaffner SF., Prasasya RD, Aharoni A, Dingwall HL, Warder B, Kamberov YG. 2021. Repeated mutation of a developmental enhancer contributed to human thermoregulatory evolution. Proc Natl Acad Sci USA 118(16): e2021722118. https://doi.org/10.1073/pnas.2021722118
Aldea D, Kamberov YG. 2022. En1 sweat we trust: how the evolution of an Engrailed 1 enhancer made humans the sweatiest ape. Temperature 9(4): 303–5. https://doi.org/10.1080/23328940.2021.2019548
Alves dos Santos MT, Smidt MP. 2011. En1 and Wnt signaling in midbrain dopaminergic neuronal development. Neural Dev 6: 23. https://doi.org/10.1186/1749-8104-6-23
Amoasii L, Sanchez-Ortiz E, Fujikawa T, Elmquist JK, Bassel-Duby R, Olson EN. 2019. NURR1 activation in skeletal muscle controls systemic energy homeostasis. Proc Natl Acad Sci USA 116(23): 11299–308. https://doi.org/10.1073/pnas.1902490116
Balthazar CH, Leite LH, Ribeiro RM, Soares DD, Coimbra CC. 2010. Effects of blockade of central dopamine D1 and D2 receptors on thermoregulation, metabolic rate and running performance. Pharmacol Rep 62(1):54-61. https://doi.org/10.1016/s1734-1140(10)70242-5
Barr WA, Pobiner B, Rowan J, Du A, Faith JT. 2022. No sustained increase in zooarchaeological evidence for carnivory after the appearance of Homo erectus. Proc Natl Acad Sci USA 119(5): e2115540119. https://doi.org/10.1073/pnas.2115540119
Bauer BA, Rogers PJ., Miller TD., Bove AA, Tyce GM. 1989. Exercise training produces changes in free and conjugated catecholamines. Med Sci Sports Exerc 21(5): 558–562.
Beasley DE, Koltz AM, Lambert JE, Fierer N, Dunn RR. 2015. The evolution of stomach acidity and its relevance to the human microbiome. PloS One 10(7): e0134116. https://doi.org/10.1371/journal.pone.0134116
Bednarik RG. 1995. Concept-mediated marking in the lower Palaeolithic. Curr Anthropol 36(4)605–34. http://dx.doi.org/10.1086/204406
Benyamin B, Pourcain B, Davis OS, Davies G, Hansell NK, Brion MJ, Kirkpatrick RM, Cents RA, Franić S, Miller MB, Haworth CM, Meaburn E, Price TS, Evans DM, Timpson N, Kemp J, Ring S, McArdle W, Medland SE, Yang J, … Visscher PM. 2014. Childhood intelligence is heritable, highly polygenic and associated with FNBP1L. Mol Psychiatry 19(2): 253–258. https://doi.org/10.1038/mp.2012.184
Best A, Kamilar JM. 2018. The evolution of eccrine sweat glands in human and nonhuman primates. J Hum Evol 117: 33–43. https://doi.org/10.1016/j.jhevol.2017.12.003
Best A, Lieberman DE, Kamilar JM. 2019. Diversity and evolution of human eccrine sweat gland density. J Therm Biol. 84: 331–8. https://doi.org/10.1016/j.jtherbio.2019.07.024
Bloemendaal M, Froböse MI, Wegman J, Zandbelt BB, van de Rest O, Cools R, Aarts E. 2018. Neuro-cognitive effects of acute tyrosine administration on reactive and proactive response inhibition in healthy older adults. eNeuro 30; 5(2): ENEURO.0035–17.2018. https://doi.org/10.1523/ENEURO.0035-17.2018
Bortz WM II. 1985. Physical exercise as an evolutionary force. J Hum Evol 14: 145–55. https://doi.org/10.1016/S0047-2484(85)80003-8
Bortz WM II, Angwin P, Mefford IN, Boarder MR, Noyce N, Barchas JD. 1981. Catecholamines, dopamine, and endorphin levels during extreme exercise. N Engl J Med 305(8): 466–7. https://doi.org/10.1016/S0047-2484(85)80003-8
Bougea A, Spantideas N, Katoulis A, Stefanis L. 2019. Levodopa-induced skin disorders in patients with Parkinson disease: a systematic literature review approach. Acta Neurol Belg 119(3): 325–36. https://doi.org/10.1007/s13760-019-01195-3
Bramble DM, Lieberman DE. 2004. Endurance running and the evolution of Homo. Nature 432(7015): 345–52. https://doi.org/10.1038/nature03052
Brodde OE. 1982. Vascular dopamine receptors: demonstration and characterization by in vitro studies. Life Sci 31(4): 289–306. https://doi.org/10.1016/0024-3205(82)90406-4
Brown PL, Bae D, Kiyatkin EA. 2007. Relationships between locomotor activation and alterations in brain temperature during selective blockade and stimulation of dopamine transmission. Neuroscience 145(1): 335–43. https://doi.org/10.1016/j.neuroscience.2006.11.028
Brozoski TJ, Brown RM, Rosvold HE, Goldman PS. 1979. Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205(4409): 929–32. https://doi.org/10.1126/science.112679
Carrier DR. 1984. The energetic paradox of human running and hominid evolution. Curr Anthropol 25: 483–95. https://doi.org/10.1086/203165
Charkoudian N. 2003. Skin blood flow in adult human thermoregulation: how it works, when it does not, and why. Mayo Clin Proc 78(5): 603–12. https://doi.org/10.4065/78.5.603
Chaudhuri KR, Todorova A, Nirenberg MJ, Parry M, Martin A, Martinez-Martin P, Rizos A, Henriksen T, Jost W, Storch A, Ebersbach G, Reichmann H, Odin P, Antonini A. 2015. A pilot prospective, multicenter observational study of dopamine agonist withdrawal syndrome in Parkinson’s Disease. Mov Disord Clin Pract 2(2): 170–4. https://doi.org/10.1002/mdc3.12141
Cho SS, Strafella AP. 2009. rTMS of the left dorsolateral prefrontal cortex modulates dopamine release in the ipsilateral anterior cingulate cortex and orbitofrontal cortex. PLoS One 4(8): e6725. https://doi.org/10.1371/journal.pone.0006725
Coon EA, Low PA. 2018. Thermoregulation in Parkinson disease. H Clin Neurol 157: 715–25. https://doi.org/10.1016/B978-0-444-64074-1.00043-4
Cox B, Lee TF. 1980. Further evidence for a physiological role for hypothalamic dopamine in thermoregulation in the rat. J Physiol 300: 7–17. https://doi.org/10.1113/jphysiol.1980.sp013147
Clark G, Henneberg M. 2021. Cognitive and behavioral modernity in Homo erectus: skull globularity and hominin brain evolution. Anthropol Rev 84(4): 467–485. https://doi.org/10.2478/anre-2021-0030
Crockett MJ. 2009. The neurochemistry of fairness: clarifying the link between serotonin and prosocial behavior. Ann N Y Acad Sci. 1167: 76–86. https://doi.org/10.1111/j.1749-6632.2009.04506.x
Cui CY, Childress V, Piao Y, Michel M, Johnson AA, Kunisada M, Ko MS, Kaestner KH, Marmorstein AD, Schlessinger D. 2012. Forkhead transcription factor FoxA1 regulates sweat secretion through Bestrophin 2 anion channel and Na-K-Cl cotransporter 1. Proc Natl Acad Sci USA. 109(4): 1199–203. https://doi.org/10.1073/pnas.1117213109
Dávid-Barrett T, Dunbar RI. 2016. Bipedality and hair loss in human evolution revisited: the impact of altitude and activity scheduling. J Hum Evol 94: 72–82. https://doi.org/10.1016/j.jhevol.2016.02.006
Davies G, Marioni RE, Liewald DC, Hill WD, Hagenaars SP, Harris SE, Ritchie SJ, Luciano M, Fawns-Ritchie C, Lyall D, Cullen B, Cox SR, Hayward C, Porteous DJ, Evans J, McIntosh AM, Gallacher J, Craddock N, Pell JP, Smith DJ, Gale CR, Deary IJ. 2016. Genome-wide association study of cognitive functions and educational attainment in UK Biobank (N=112 151). Mol Psychiatry 21(6): 758–67. https://doi.org/10.1038/mp.2016.45
Daw ND, Kakade S, Dayan P. 2002. Opponent interactions between serotonin and dopamine. Neural Netw 15(4–6): 603–16. https://doi.org/10.1016/s0893-6080(02)00052-7
Dean MC, Cole TJ. 2013. Human life history evolution explains dissociation between the timing of tooth eruption and peak rates of root growth. PloS One 8(1): e54534. https://doi.org/10.1371/journal.pone.0054534
DeLouize AM, Coolidge FL, Wynn T. 2017. Dopaminergic systems expansion and the advent of Homo erectus. Quat Int 427(B): 245–52. https://doi.org/10.1016/j.quaint.2015.10.123
D’Esposito M, Postle BR. 2015. The cognitive neuroscience of working memory. Annu Rev Psychol 66: 115–42. https://doi.org/10.1146/annurev-psych-010814-015031
Desmond MA, Sobiecki JG, Jaworski M, Płudowski P, Antoniewicz J, Shirley MK, Eaton S, Książyk J, Cortina-Borja M, De Stavola B, Fewtrell M, Wells JCK. 2021. Growth, body composition, and cardiovascular and nutritional risk of 5- to 10-y-old children consuming vegetarian, vegan, or omnivore diets. Am J Clin Nutr 113(6): 1565–77. https://doi.org/10.1093/ajcn/nqaa445
DeYoung CG, Cicchetti D, Rogosch FA, Gray JR, Eastman M, Grigorenko EL 2011. Sources of cognitive exploration: Genetic variation in the prefrontal dopamine system predicts openness/intellect. J Res Pers. 45(4): 364–371. https://doi.org/10.1016/j.jrp.2011.04.002
Dhugga A, Henneberg M, Kumaratilake JS. 2014. Variation of human hairiness: a possible adaptation to solar radiation and melanin. Anthropol Rev 77(2): 219–32. https://doi.org/10.2478/anre-2014-0017
Di Domenico D, Mapelli L. 2023. Dopaminergic modulation of prefrontal cortex inhibition. Biomedicines 11(5): 1276. https://doi.org/10.3390/biomedicines11051276
Doll HM, Risgaard RD, Thurston H, Chen RJ, Sousa AM. 2024. Evolutionary innovations in the primate dopaminergic system. Curr Opin Genet Dev 88:102236. https://doi.org/10.1016/j.gde.2024.102236
Ferraro JV, Plummer TW, Pobiner BL, Oliver JS, Bishop LC, Braun DR, Ditchfield PW, Seaman JW 3rd, Binetti KM, Seaman JW Jr, Hertel F, Potts R. 2013. Earliest archaeological evidence of persistent hominin carnivory. PLoS One 8(4): e62174. https://doi.org/10.1371/journal.pone.0062174
Ferri AL, Lin W, Mavromatakis YE, Wang JC, Sasaki H, Whitsett JA, Ang SL. 2007. Foxa1 and Foxa2 regulate multiple phases of midbrain dopaminergic neuron development in a dosage-dependent manner. Development 134(15): 2761–9. https://doi.org/10.1242/dev.000141
Fuertinger S, Zinn JC, Sharan AD, Hamzei-Sichani F, Simonyan K. 2018. Dopamine drives left-hemispheric lateralization of neural networks during human speech. J Comp Neurol 526(5): 920–31. https://doi.org/10.1002/cne.24375
Garcia-Garcia M, Barceló F, Clemente IC, Escera C. 2010. The role of the dopamine transporter DAT1 genotype on the neural correlates of cognitive flexibility. Eur J Neurosci 31(4): 754–60. https://doi.org/10.1111/j.1460-9568.2010.07102.x
Goldman PS, Rosvold HE. 1972. The effects of selective caudate lesions in infant and juvenile Rhesus monkeys. Brain Res 43(1): 53–66. https://doi.org/10.1016/0006-8993(72)90274-0
Grazioplene RG, G Ryman S, Gray JR, Rustichini A, Jung RE, DeYoung CG. 2015. Subcortical intelligence: caudate volume predicts IQ in healthy adults. Hum Brain Mapp 36(4): 1407–16. https://doi.org/10.1002/hbm.22710
Hart G, Burton TJ, Balleine, BW 2024. What role does striatal dopamine play in goal-directed action? Neuroscience 546: 20–32. https://doi.org/10.1016/j.neuroscience.2024.03.020
Hase A, Jung SE, aan het Rot M. 2015. Behavioral and cognitive effects of tyrosine intake in healthy human adults. Pharmacol Biochem Behav 133: 1–6. https://doi.org/10.1016/j.pbb.2015.03.008
Henneberg M, Sarafis V. 1998. Human adaptations to meat eating. Hum Evol 13: 229–34. http://dx.doi.org/10.1007/bf02436507
Hirter KN, Miller EN, Stimpson CD, Phillips KA, Hopkins WD, Hof PR, Sherwood CC, Lovejoy CO, Raghanti MA. 2021. The nucleus accumbens and ventral pallidum exhibit greater dopaminergic innervation in humans compared to other primates. Brain Struct Funct 226(6): 1909–23. https://doi.org/10.1007/s00429-021-02300-0
Hirschbeck A, Leao DS, Wagner E, Hasan A, Roeh A. 2022. Psychiatric medication and physical performance parameters – Are there implications for treatment? Front Psychiatry 13:985983. https://doi.org/10.3389/fpsyt.2022.985983
Hoberg EP, Alkire NL, de Queiroz A, Jones A. 2001. Out of Africa: origins of the Taenia tapeworms in humans. Proc Biol Sci 268(1469): 781–7. https://doi.org/10.1098/rspb.2000.1579
Horackova H, Karahoda R, Vachalova V, Turkova H, Abad C, Staud F. 2022. Functional characterization of dopamine and norepinephrine transport across the apical and basal plasma membranes of the human placental syncytiotrophoblast. Sci Rep 12(1): 11603. https://doi.org/10.1038/s41598-022-15790-7
Ikemoto K, Kitahama K, Jouvet A, Arai R, Nishimura A, Nishi K, Nagatsu I. 1997. Demonstration of L-dopa decarboxylating neurons specific to human striatum. Neurosci Lett 29;232(2): 111–4. https://doi.org/10.1016/s0304-3940(97)00587-9
Jablonski NG. 2021. The evolution of human skin pigmentation involved the interactions of genetic, environmental, and cultural variables. Pigment Cell Melanoma Res 34(4): 707–29. https://doi.org/10.1111/pcmr.12976
Jaouen K, Beasley M, Schoeninger M, Hublin JJ, Richards MP. 2016. Zinc isotope ratios of bones and teeth as new dietary indicators: results from a modern food web (Koobi Fora, Kenya). Sci Rep 6: 26281. https://doi.org/10.1038/srep26281
Jaouen K, Villalba-Mouco V, Smith GM, Trost M, Leichliter J, Lüdecke T, Méjean P, Mandrou S, Chmeleff J, Guiserix D, Bourgon N, Blasco F, Mendes Cardoso J, Duquenoy C, Moubtahij Z, Salazar Garcia DC, Richards M, Tütken T, Hublin JJ, Utrilla P, Montes L. 2022. A Neandertal dietary conundrum: insights provided by tooth enamel Zn isotopes from Gabasa, Spain. Proc Natl Acad Sci USA 119(43): e2109315119. https://doi.org/10.1073/pnas.2109315119
Kamberov YG, Guhan SM, DeMarchis A, Jiang J, Wright SS, Morgan BA, Sabeti PC, Tabin CJ, Lieberman DE. 2018. Comparative evidence for the independent evolution of hair and sweat gland traits in primates. J Hum Evol 125: 99–105. https://doi.org/10.1016/j.jhevol.2018.10.008
Kaminski JA, Schlagenhauf F, Rapp M, Awasthi S, Ruggeri B, Deserno L, Banaschewski T, Bokde ALW, Bromberg U, Büchel C, Quinlan EB, Desrivières S, Flor H, Frouin V, Garavan H, Gowland P, Ittermann B, Martinot JL, Martinot MP, Nees F, Orfanos DP, Paus T, Poustka L, Smolka MN, Fröhner JH, Walter H, Whelan R, Ripke S, Schumann G, Heinz A; IMAGEN consortium. 2018. Epigenetic variance in dopamine D2 receptor: a marker of IQ malleability? Transl Psychiatry 8(1): 169. https://doi.org/10.1038/s41398-018-0222-7
Khalaf K, Brook AH, Smith RN. 2022. Genetic, epigenetic and environmental factors influence the phenotype of tooth number, size and shape: anterior maxillary supernumeraries and the morphology of mandibular incisors. Genes (Basel) (12): 2232. https://doi.org/10.3390/genes13122232
Kling A, Tucker TJ. 1967. Effects of combined lesions of frontal granular cortex and caudate nucleus in the neonatal monkey. Brain Res 6(3): 428–39. https://doi.org/10.1016/0006-8993(67)90056-x
Ko JH, Monchi O, Ptito A, Bloomfield P, Houle S, Strafella AP. 2008. Theta burst stimulation-induced inhibition of dorsolateral prefrontal cortex reveals hemispheric asymmetry in striatal dopamine release during a set-shifting task: a TMS-[(11)C]raclopride PET study. Eur J Neurosci 28(10): 2147–55. https://doi.org/10.1111/j.1460-9568.2008.06501.x
Kühn S, Düzel S, Colzato L, Norman K, Gallinat J, Brandmaier AM, Lindenberger U, Widaman KF. 2019. Food for thought: association between dietary tyrosine and cognitive performance in younger and older adults. Psychol Res. 83(6): 1097–1106. https://doi.org/10.1007/s00426-017-0957-4
Langan EA, Lisztes E, Bíró T, Funk W, Kloepper JE, Griffiths CE, Paus R. 2013. Dopamine is a novel, direct inducer of catagen in human scalp hair follicles in vitro. Br J Dermatol. 168(3): 520–5. https://doi.org/10.1111/bjd.12113
Larisch R, Meyer W, Klimke A, Kehren F, Vosberg H, Müller-Gärtner HW. 1998. Left-right asymmetry of striatal dopamine D2 receptors. Nucl Med Commun 19(8): 781–7. https://doi.org/10.1097/00006231-199808000-00009
Larsen T, Fernandes R, Wang YV, Roberts P. 2022. Reconstructing hominin diets with stable isotope analysis of amino acids: new perspectives and future directions. Bioscience 72(7): 618–637. https://doi.org/10.1093/biosci/biac028
Leclercq M, Gimenes G, Maintenant C, Clerc J. 2023. Goal choice in preschoolers is influenced by context, cognitive flexibility, and metacognition. Front Psychol 13: 1063566. https://doi.org/10.3389/fpsyg.2022.1063566
Lee TF, Mora F, Myers RD. 1985. Dopamine and thermoregulation: an evaluation with special reference to dopaminergic pathways. Neurosci Biobehav Rev 9(4): 589–98. https://doi.org/10.1016/0149-7634(85)90005-3
Lee J, Ryu HJ, Hwang SY, Koh SB. 2024. Hair loss: a well-known yet understudied symptom in Parkinson’s Disease patients during dopaminergic therapy. J Mov Disord 17(1): 47–54. https://doi.org/10.14802/jmd.23088.
Le Pen G, Sonnier L, Hartmann A, Bizot JC, Trovero F, Krebs MO, Prochiantz A. 2008. Progressive loss of dopaminergic neurons in the ventral midbrain of adult mice heterozygote for Engrailed1: a new genetic model for Parkinson’s disease? Parkinsonism Relat Disord 14()2: S107–11. https://doi.org/10.1016/j.parkreldis.2008.04.007
Lhommée E, Batir A, Quesada JL, Ardouin C, Fraix V, Seigneuret E, Chabardès S, Benabid AL, Pollak P, Krack P. 2014. Dopamine and the biology of creativity: lessons from Parkinson’s disease. Front Neurol 5: 55. https://doi.org/10.3389/fneur.2014.00055
Lieberman DE. 2015. Human locomotion and heat loss: an evolutionary perspective. Compr Physiol 5(1): 99–117. https://doi.org/10.1002/cphy.c140011
Linares C, Martinez-Martin P, Rodríguez-Blázquez C, Forjaz MJ, Carmona R, Díaz J. 2016. Effect of heat waves on morbidity and mortality due to Parkinson’s disease in Madrid: A time-series analysis. Environ Int 89–90: 1–6. https://doi.org/10.1016/j.envint.2016.01.017
Luciana M, Collins PF, Depue, RA. 1998. Opposing roles for dopamine and serotonin in the modulation of human spatial working memory functions. Cereb Cortex 8(3): 218–26. https://doi.org/10.1093/cercor/8.3.218
Lucock MD. 2023. The evolution of human skin pigmentation: a changing medley of vitamins, genetic variability, and UV radiation during human expansion. Am J Biol Anthropol 180(2): 252–71. https://doi.org/10.1002/ajpa.24564
Ma S, Skarica M, Li Q, Xu C, Risgaard RD, Tebbenkamp ATN, Mato-Blanco X, Kovner R, Krsnik Ž, de Martin X, Luria V, Martí-Pérez X, Liang D, Karger A, Schmidt DK, Gomez-Sanchez Z, Qi C, Gobeske KT, Pochareddy S, Debnath A, Hottman CJ, Spurrier J, Teo L, Boghdadi AG, Homman-Ludiye J, Ely JJ, Daadi EW, Mi D, Daadi M, Marín O, Hof PR, Rasin MR, Bourne J, Sherwood CC, Santpere G, Girgenti MJ, Strittmatter SM, Sousa AMM, Sestan N. 2022. Molecular and cellular evolution of the primate dorsolateral prefrontal cortex. Science 377(6614): eabo7257. https://doi.org/10.1126/science.abo7257.
Maranduca MA, Branisteanu D, Serban DN, Branisteanu DC, Stoleriu G, Manolache N, Serban IL. 2019. Synthesis and physiological implications of melanic pigments. Oncol Lett 17(5): 4183–7. https://doi.org/10.3892/ol.2019.10071
Marques PR, Spencer RL, Burks TF, McDougal JN. 1984. Behavioral thermoregulation, core temperature, and motor activity: simultaneous quantitative assessment in rats after dopamine and prostaglandin E1. Behav Neurosci 98(5): 858–67. https://doi.org/10.1037//0735-7044.98.5.858
Matsuda W, Furuta T, Nakamura KC, Hioki H, Fujiyama F, Arai R, Kaneko T. 2009. Single nigrostriatal dopaminergic neurons form widely spread and highly dense axonal arborizations in the neostriatum. J Neurosci 29(2): 444–53. https://doi.org/10.1523/JNEUROSCI.4029-08.2009
Matzel LD, Sauce B. 2023. A multi-faceted role of dual-state dopamine signaling in working memory, attentional control, and intelligence. Front Behav Neurosci 17: 1060786. https://doi.org/10.3389/fnbeh.2023.1060786
Mitchell JM, Weinstein D, Vega T, Kayser AS. 2018. Dopamine, time perception, and future time perspective. Psychopharmacology (Berl) 235(10): 2783–93. https://doi.org/10.1007/s00213-018-4971-z
Montgomery AJ, McTavish SF, Cowen PJ, Grasby PM. 2003. Reduction of brain dopamine concentration with dietary tyrosine plus phenylalanine depletion: an [11C]raclopride PET study. Am J Psychiatry 160(10): 1887–9. https://doi.org/10.1176/appi.ajp.160.10.1887
Mosites E, Aol G, Otiang E, Bigogo G, Munyua P, Montgomery JM, Neuhouser ML, Palmer GH, Thumbi SM. 2017. Child height gain is associated with consumption of animal-source foods in livestock-owning households in Western Kenya. Public Health Nutr 20(2): 336–45. https://doi.org/10.1017/S136898001600210X
Newman EJ, Grosset DG, Kennedy PG. 2009. The parkinsonism-hyperpyrexia syndrome. Neurocrit Care 10(1): 136–40. https://doi.org/10.1007/s12028-008-9125-4.
Nordströma U, Beauvais G, Ghosh A, Pulikkaparambil Sasidharan BC, Lundblad M, Fuchs J, Joshi RL, Lipton JW, Roholt A, Medicetty S, Feinstein TN, Steiner JA, Escobar Galvis ML, Prochiantz A, Brundin P. 2015. Progressive nigrostriatal terminal dysfunction and degeneration in the engrailed1 heterozygous mouse model of Parkinson’s disease. Neurobiol Dis 73: 70–82. https://doi.org/10.1016/j.nbd.2014.09.012
Nouri N, Awatramani R. 2017. A novel floor plate boundary defined by adjacent En1 and Dbx1 microdomains distinguishes midbrain dopamine and hypothalamic neurons. Development 144(5): 916–27. https://doi.org/10.1242/dev.144949
Olvera-Cortés ME, Anguiano-Rodríguez P, López-Vázquez MA, Alfaro JM. 2008. Serotonin/dopamine interaction in learning. Prog Brain Res 172: 567–602. https://doi.org/10.1016/S0079-6123(08)00927-8
Palmiero M, Fusi G, Crepaldi M, Borsa VM, Rusconi ML. 2022. Divergent thinking and the core executive functions: a state-of-the-art review. Cogn Process 23(3): 341–366. https://doi.org/10.1007/s10339-022-01091-4
Pietschnig J, Penke L, Wicherts JM, Zeiler M, Voracek M. 2015. Meta-analysis of associations between human brain volume and intelligence differences: how strong are they and what do they mean? Neurosci Biobehav Rev 57: 411–32. https://doi.org/10.1016/j.neubiorev.2015.09.017
Pobiner BL. 2020. The zooarchaeology and paleoecology of early hominin scavenging. Evol Anthropol 29(2): 68–82. https://doi.org/10.1002/evan.2182
Previc FH. 1991. A general theory concerning the prenatal origins of cerebral lateralization in humans. Psychol Rev 98(3): 299–334. https://doi.org/10.1037/0033-295x.98.3.299
Previc FH. 1999. Dopamine and the origins of human intelligence. Brain Cogn 41(3): 299–350. https://doi.org/10.1006/brcg.1999.1129
Previc FH. 2009. The Dopaminergic Mind in Human Evolution and History. New York: Cambridge University Press.
Quinn G. 2012. Normal genetic variation of the human foot: part 1: the paradox of normal anatomical alignment in an evolutionary epigenetic context. J Am Podiatr Med Assoc 102(1): 64–70. https://doi.org/10.7547/1020064
Raghanti MA, Edler MK, Stephenson AR, Wilson LJ, Hopkins WD, Ely JJ, Erwin JM, Jacobs B, Hof PR, Sherwood CC. 2016. Human-specific increase of dopaminergic innervation in a striatal region associated with speech and language: a comparative analysis of the primate basal ganglia. J Comp Neurol 524(10): 2117–29. https://doi.org/10.1002/cne.23937
Raghanti MA, Edler MK, Stephenson AR, Munger EL, Jacobs B, Hof PR, Sherwood CC, Holloway RL, Lovejoy CO. 2018. A neurochemical hypothesis for the origin of hominids. Proc Natl Acad Sci USA 115(6): E1108-E1116. https://doi.org/10.1073/pnas.1719666115
Raghanti MA, Spocter MA, Stimpson CD, Erwin JM, Bonar CJ, Allman JM, Hof PR, Sherwood CC. 2009. Species-specific distributions of tyrosine hydroxylase-immunoreactive neurons in the prefrontal cortex of anthropoid primates. Neuroscience 158(4): 1551–9. https://doi.org/10.1016/j.neuroscience.2008.10.058
Ranganath A, Jacob SN. 2016. Doping the mind: dopaminergic modulation of prefrontal cortical cognition. Neuroscientist 22(6): 593–603. https://doi.org/10.1177/1073858415602850
Rusz J, Dusek P, Tykalova T, Novotny M, Illner V, Simek M, Kouba T, Kryze P, Zogala D, Ruzicka E, Sousa M, Jorge A, Nef T, Krack P. 2024. Is speech function lateralised in the basal ganglia? Evidence from de novo Parkinson’s disease. J Neurol Neurosurg Psychiatry jnnp-2024-334297. https://doi.org/10.1136/jnnp-2024-334297
Ruxton GD, Wilkinson DM. 2011. Avoidance of overheating and selection for both hair loss and bipedality in hominins. Proc Natl Acad Sci USA 108(52): 20965–9. https://doi.org/10.1073/pnas.1113915108
Saniotis A, Grantham JP, Kumaratilake JS, Henneberg M, Mohammadi K. 2021. Going beyond brain size: An evolutionary overview of serotonergic regulation in human higher cortical functions. Anthropologie 59(1): 101–6. https://doi.org/10.26720/anthro.20.08.1
Scott IM, Boulant JA. 1984. Dopamine effects on thermosensitive neurons in hypothalamic tissue slices. Brain Res 306(1–2): 157–163. https://doi.org/10.1016/0006-8993(84)90364-0
Shibasaki M, Crandall CG. 2010. Mechanisms and controllers of eccrine sweating in humans. Front Biosci (Schol Ed) 2(2): 685–96. https://doi.org/10.2741/s94
Simon HH, Thuret S, Alberi L. 2004. Midbrain dopaminergic neurons: control of their cell fate by the engrailed transcription factors. Cell Tissue Res 318(1): 53–61. https://doi.org/10.1007/s00441-004-0973-8
Simonyan K, Herscovitch P, Horwitz B. 2013. Speech-induced striatal dopamine release is left lateralized and coupled to functional striatal circuits in healthy humans: a combined PET, fMRI and DTI study. Neuroimage 70: 21–32. https://doi.org/10.1016/j.neuroimage.2012.12.042
Smith JW, Bello ML, Price FG. 2021. A case-series observation of sweat rate variability in endurance-trained athletes. Nutrients 13(6): 1807. https://doi.org/10.3390/nu13061807
Sniekers S, Stringer S, Watanabe K, Jansen PR, Coleman JRI, Krapohl E, Taskesen E, Hammerschlag AR, Okbay A, Zabaneh D, Amin N, Breen G, Cesarini D, Chabris CF, Iacono WG, Ikram MA, Johannesson M, Koellinger P, Lee JJ, Magnusson PKE, McGue M, Miller MB, Ollier WER, Payton A, Pendleton N, Plomin R, Rietveld CA, Tiemeier H, van Duijn CM, Posthuma D. 2017. Genome-wide association meta-analysis of 78,308 individuals identifies new loci and genes influencing human intelligence. Nat Genet 49(10): 1558. https://doi.org/10.1038/ng1017-1558c. Erratum for: Nat Genet. 2017 49(7): 1107–1112. https://doi.org/10.1038/ng.3869
Sousa AMM, Zhu Y, Raghanti MA, Kitchen RR, Onorati M, Tebbenkamp ATN, Stutz B, Meyer KA, Li M, Kawasawa YI, Liu F, Perez RG, Mele M, Carvalho T, Skarica M, Gulden FO, Pletikos M, Shibata A, Stephenson AR, Edler MK, Ely JJ, Elsworth JD, Horvath TL, Hof PR, Hyde TM, Kleinman JE, Weinberger DR, Reimers M, Lifton RP, Mane SM, Noonan JP, State MW, Lein ES, Knowles JA, Marques-Bonet T, Sherwood CC, Gerstein MB, Sestan N. 2017. Molecular and cellular reorganization of neural circuits in the human lineage. Science 358(6366): 1027–32. https://doi.org/10.1126/science.aan3456
Stanley SM. 1995. Climatic forcing and the origin of the human genus. In: National Research Council (US) Panel on Effects of Past Global Change on Life. Washington, DC: National Academies Press, 233–43.
Suzuki T. 1981. How great will the stature of Japanese eventually become? J Hum Ergol (Tokyo) 10(1): 13–24.
Takeuchi H, Taki Y, Sekiguchi A, Nouchi R, Kotozaki Y, Nakagawa S, Miyauchi CM, Iizuka K, Yokoyama R, Shinada T, Yamamoto Y, Hanawa S, Araki T, Hashizume H. 2014. Creativity measured by divergent thinking is associated with two axes of autistic characteristics. Front Psychol 5: 921. https://doi.org/10.3389/fpsyg.2014.00921
Taki Y, Hashizume H, Sassa Y, Takeuchi H, Asano M, Asano K, Kotozaki Y, Nouchi R, Wu K, Fukuda H, Kawashima R. 2012. Correlation among body height, intelligence, and brain gray matter volume in healthy children. Neuroimage 59(2): 1023–7. https://doi.org/10.1016/j.neuroimage.2011.08.092
Tam CS, Johnson WD, Rood J, Heaton AL, Greenway FL. 2020. Increased human growth hormone after oral consumption of an amino acid supplement: results of a randomized, placebo-controlled, double-blind, crossover study in healthy subjects. Am J Ther. 27(4): e333–e337. https://doi.org/10.1097/MJT.0000000000000893
Taylor KM, Giersch GEW, Caldwell AR, Epstein Y, Charkoudian N. 2024. Relation of body surface area-to-mass ratio to risk of exertional heat stroke in healthy men and women. J Appl Physiol (1985). 136(3): 549–54. https://doi.org/10.1152/japplphysiol.00597.2023
Tilkens MJ, Wall-Scheffler C, Weaver TD, Steudel-Numbers K. 2007. The effects of body proportions on thermoregulation: an experimental assessment of Allen’s rule. J Hum Evol 53(3): 286–91. https://doi.org/10.1016/j.jhevol.2007.04.005
Tong J, Hornykiewicz O, Kish SJ. 2006. Inverse relationship between brain noradrenaline level and dopamine loss in Parkinson disease: a possible neuroprotective role for noradrenaline. Arch Neurol 63(12):1724-8. https://doi.org/10.1001/archneur.63.12.1724
Tormoehlen LM, Rusyniak DE. 2018. Neuroleptic malignant syndrome and serotonin syndrome. Handb Clin Neurol 157: 663–75. https://doi.org/10.1016/B978-0-444-64074-1.00039-2
Toups MA, Kitchen A, Light JE, Reed DL. 2011. Origin of clothing lice indicates early clothing use by anatomically modern humans in Africa. Mol Biol Evol 28(1): 29–32. https://doi.org/10.1093/molbev/msq234
Tyler J, Podaras M, Richardson B, Roeder N, Hammond N, Hamilton J, Blum K, Gold M, Baron DA, Thanos PK. 2023. High intensity interval training exercise increases dopamine D2 levels and modulates brain dopamine signaling. Front Public Health 11: 1257629. https://doi.org/10.3389/fpubh.2023.1257629
Wang L, Yin H, Di Y, Liu Y, Liu J. 2016. Human local and total heat losses in different temperature. Physiol Behav 157: 270–6. https://doi.org/10.1016/j.physbeh.2016.02.018
Weydt P, Dupuis L, Petersen Å. 2018. Thermoregulatory disorders in Huntington disease. Handb Clin Neurol 157: 761–75. https://doi.org/10.1016/B978-0-444-64074-1.00047-1
Wheeler PE. 1985. The loss of functional body hair in man: the influence of thermal environment, body form and bipedality. J Hum Evol 14: 23–8. https://doi.org/10.1016/S0047-2484(85)80091-9
Will M, Pablos A, Stock JT. 2017. Long-term patterns of body mass and stature evolution within the hominin lineage. R Soc Open Sci 4(11): 171339. https://doi.org/10.1098/rsos.171339
Yuan T, Ying J, Jin L, Li C, Gui S, Li Z, Wang R, Zuo Z, Zhang Y. 2020. The role of serum growth hormone and insulin-like growth factor-1 in adult humans brain morphology. Aging (Albany NY). 12(2): 1377–96. https://doi: 10.18632/aging.102688. Erratum in: Aging (Albany NY). 2021 Sep 29;13(18): 22623–22624. https://doi.org/10.18632/aging.203601
Zabelina DL, Colzato L, Beeman M, Hommel B. 2016. Dopamine and the creative mind: individual differences in creativity are predicted by interactions between dopamine genes DAT and COMT. PLoS One 11(1): e0146768. https://doi.org/10.1371/journal.pone.0146768
Zheng X, Hasegawa H. 2016. Central dopaminergic neurotransmission plays an important role in thermoregulation and performance during endurance exercise. Eur J Sport Sci 16(7): 818–28. https://doi.org/10.1080/17461391.2015.1111938
Zink KD, Lieberman DE. 2016. Impact of meat and Lower Palaeolithic food processing techniques on chewing in humans. Nature 531(7595): 500–3. https://doi.org/10.1038/nature16990
Final information
Acknowledgements
The author wishes to acknowledge the assistance of Dr. Britt Bousman in the conceptual stage of this paper.
Conflict of interest statement
There are no conflicts of interest associated with this paper.
Author contribution
Sole author responsible for conceptualization, literature synthesis, writing and editing of the manuscript.
Ethics statement
Not applicable.
Data availability statement
Not applicable.
Financial disclosure
No outside funding sources were involved.
Corresponding author
Fred H. Previc, Department of Psychology, The University of Texas at San Antonio, One UTSA Circle, San Antonio TX 78249, USA, e-mail: fprevic@gmail.com