Predicting Prosociality in Primates: Socio-Ecological Influences and a Framework of Inter-Brain Neural Synchronization
 https://orcid.org/0000-0003-4078-6178
https://orcid.org/0000-0003-4078-6178
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
 https://orcid.org/0000-0003-1941-2286
https://orcid.org/0000-0003-1941-2286
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
Institute for Evolutionary Medicine, University of Zurich, Winterthurerstrasse 190, 8057 Zürich, Switzerland
 https://orcid.org/0000-0002-6228-6150
https://orcid.org/0000-0002-6228-6150
School of Psychology, University of Adelaide, Adelaide, South Australia, 5005, Australia
 https://orcid.org/0000-0001-9317-6352
https://orcid.org/0000-0001-9317-6352
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
Allied Health & Human Performance, University of South Australia, Adelaide, South Australia, 5001, Australia
 https://orcid.org/0000-0002-9708-6220
https://orcid.org/0000-0002-9708-6220
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
 https://orcid.org/0000-0002-6229-1064
https://orcid.org/0000-0002-6229-1064
School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia
School of Nursing and Midwifery, Western Sydney University, Penrith, New South Wales, 2751, Australia
Abstract. Prosocial co-operation is critical for evolution and survival on Earth and has crucially shaped the development of Homo sapiens. Inter-brain neural synchronization (IBNS) has been shown to enhance prosocial co-operation in mammals and avians. The selection pressures which led to the development of IBNS throughout primate evolution are currently unknown. This paper aims to expand the understanding of IBNS in non-human primates by reviewing the literature on various primate populations that display prosocial behaviors that could correlate with IBNS. Binary logit modelling using machine learning methods was applied to social, ecological, morphological, and biological (SEMB) variables correlated with prosocial behaviors to obtain probabilities of prosociality. Our results suggest that select SEMB variables such as daily socialisation, food-sharing and hierarchy structure are strong predictors of prosocial behaviors in primates. We provide a framework that offers testable hypotheses for the existence of IBNS in primates based on the correlations between SEMB variables and prosocial behaviors. We also offer ideas of the ecological/behavioral forces that may correlate with neural activation patterns of primate IBNS. Through comparison to Homo sapiens models, these findings suggest IBNS in primates may exist beyond cercopithecids and may be evoked by similar socio-ecological contexts. However, some key neurological distinctions between the two groups exist, influencing which distinct patterns of behavior may evoke IBNS (relative to their socio-ecological context).
Keywords: prosociality, comparative behavior, inter-brain neural synchronization, neural systems, machine learning
Introduction
When analysing the evolution of life on Earth, a common trend prevails – co-operation can become a vital key to reproductive success. From the smallest amoeba to Homo sapiens, co-operation birthed longevity (Kreider et al. 2022; Zhu et al. 2023). In retrospect, if co-operation in the Hominidae clade disappeared some 3 million years ago, H. sapiens could not have evolved. Our hominin ancestors needed co-operation as a survival tool to combat harsh environments that posed risks to their small population numbers (Townsend et al. 2023). Evolution of the Homo genus was dependent on the cooperative behaviors of our ancestors, who evolved from Miocene primates. Most extant primates still rely on cooperation to survive (Boyd and Richerson 2009).
From this point forward, when we refer to primates we exclude the tribe Hominini of which H. sapiens and their immediate ancestors are members. Primates are faced with a myriad of unique threats to their survival in various environments including predators, terrain, food security, and so on. However, using co-operation as a tool to protect against these forces is a common strategy used in prosocial behaviors such as unified defence against predators or foraging as a group to increase food collection (Garber 1987; Treves 2000; Boinski and Garber 2001). Though co-operative behaviors can be quite varied, all can be collectively classified as prosocial behaviors as they benefit the group instead of (or as well as) the individual primate (De Waal and Suchak 2010). The nature of primate prosocial behavior is different when compared to H. sapiens. Prosociality in H. sapiens is distinguished by a stronger empathic drive that employs a wider range of enforcement mechanisms, allowing for more stable cooperation to form in larger groups of unrelated individuals (Melis and Semmann 2010). This enabled H. sapiens to flourish in the late Pleistocene (Sterelny 2021; Townsend et al. 2023), leading to the large population size increase seen today. How then do extant non-human primates sustain prosocial behavior without the usage of complex enforcement mechanisms cognate to H. sapiens? One answer to this question may be found in inter-brain neural synchronization (IBNS).
Inter-brain neural synchronization is an unconscious mechanism; where neural oscillatory activity is synchronized between people, so that recorded neural behavior across brain regions and bandwidths associated with prosocial tasks is identical (Valencia and Froese 2020; Lotter et al. 2023). Observational and experimental studies in mammals (including H. sapiens) describe IBNS as a neural behavior pattern evoked by prosocial behaviors such as alloparenting, physical co-ordination and group tasks serving a common goal (Tseng et al. 2018; Kingsbury et al. 2019; Rose et al. 2021; Zhang et al. 2022; Ogawa and Shimada. 2023). A distinction between mirror neuron activity and IBNS must be made here, with implications to identify IBNS as a facilitative process for the achievement of social goals. As mirror neurons are grounded in motor-based behavior (inter-personal motor synchrony) it is difficult to generalise their synchronization effect to abstract situations between people (e.g. conversational turn-taking) (Hasson and Frith 2016). However, IBNS does result in cortical synchronization between areas representing abstract processes and even correlates to enhancements of abstract cognition (e.g. shared intentionality) (Fishburn et al. 2018; Gvirtz-Provolovski and Perlmutter 2021). Thus, IBNS is distinct from mirror neurons as there is no proof that it is a static process; it is more likely that IBNS fluctuates between synchronized and desynchronized periods (Froese et al. 2024). Wang et al. (2025) demonstrate that by separating the effects of motor synchronization (mirroring) from genuine IBNS, brain activity synchronized specifically to abstract cognition (e.g. emotion) can be isolated. Therefore, IBNS is characterized by the synchronization of neural activity underpinning abstract cognition. Prosociality often requires abstract cognition as it involves understanding and acting on shared goals, beliefs, or expectations; considering the perspectives or needs of others (Houwelingen and Dijke 2023). Considering this, IBNS is more than a computed correlation of synchronized brain regions, as previous hyper-scanning studies strongly indicate that prosociality mediates the IBNS process.
In H. sapiens, IBNS is a typical response associated with prosocial activity requiring shared intention and is suggested to enhance co-operation and cohesion in prosocial tasks, especially when compared to the same scenarios where IBNS is weak or not evoked between people (Rennung and Goritz 2016; Hu et al. 2017; Lu et al. 2023; Wass et al. 2020; Wang et al. 2025). Considering these benefits, IBNS could play a crucial, facilitative role in the co-operation of primates where there is an absence of similar levels of enforcement mechanisms which motivate stable prosocial behavior in H. sapiens (Koski and Sterck 2009; Melis and Semmann 2010).
Little information on IBNS in primates exists (Ramakrishnan et al. 2015; Tseng et al. 2018). It should be noted that the current IBNS literature in primates is based solely on macaques (cercopithecids). None of these studies examine IBNS elicited through natural engagement in prosocial behaviors, and thus overlook aspects of the socio-ecological contexts that select for such behaviors. In this paper, it is proposed that our IBNS framework in primates will help researchers identify populations where IBNS may be easily observed. These assumptions are based on predictive correlations using the overarching socio-ecological contexts of primates, which are likely to elicit displays of prosociality. The proposed framework could also be made falsifiable by fieldwork that reports consistent prosocial behavior in primates that does not evoke IBNS. Both outcomes would provide distinct insight into the evolution of IBNS in Homo sapiens.
A study of IBNS in Macaca mulatta dyads performing a socio-motor task where one member came into the vicinity of a food item while the other observed, showed that M. mulatta exhibited IBNS that was dependent on specific aspects of social interaction and cognition (Tseng et al. 2018). Ecological conditions such as the food reward (and its location) and the presence of conspecifics influenced the IBNS response, underpinned possibly by dopaminergic reward-response pathways critical to social learning and adaptation in primates (Murray et al. 2011; Beeler and Dryer. 2019; Wise 2020). Tseng et al. (2018) present a possible relationship between primate IBNS and specific stimuli/co-operative behaviors influencing IBNS activation patterns (with socio-ecological forces potentially acting as a mediating factor). An identical framework for IBNS is found in H. sapiens (Valencia and Froese 2020). However, this paper offers a broader hypothetical framework for primate IBNS that derives from analysis of potential differences in the elicitation and manifestation of this function across a wide range of primate species. We suggest that future research based on hypotheses proposed here may lead to the verification of IBNS in a wide range of primates.
Inter-Brain Neural Synchronization in animals extends beyond the primates; bats, rats and birds also demonstrate IBNS concomitant with increased cooperation and social adaptation (Hoffman et al. 2019; Kingsbury et al. 2019; Zhang and Yartsev 2019; Rose et al. 2021; Zhang et al.,2022). Notably, no modern reptile has been documented to exhibit IBNS, though social complexity influenced by ecological variables has been recorded (Brattstrom 1974). Though reptiles are stereotypically labelled as non-social, both current and ancient reptilians present valid cases of prosocial behavior (Doody et al. 2012; Titus et al. 2021). Therefore, their potential for IBNS should not be overlooked, which raises two important considerations: 1. Ecology influences the emergence of prosociality and 2. The absence of a prosocial behavior does not preclude any species from having a latent aptitude for IBNS. Considering this, we cannot say whether IBNS may exist as a homoplasy or homology in evolution (in part due to the absence of IBNS research in reptiles and other non-mammalian vertebrates such as fish). So far IBNS has been recorded in some animals of mammalian ancestry; within this lineage there also exist animals who display little prosociality. The naturally solitary Pongo abelli is a prime example, though they are still capable of prosocial behavior and even display conflict resolution skills under crowded living conditions with conspecifics (an example of prosocial behavior influenced by socio-ecological forces) (Kopp and Liebal 2018). This fact could further the argument of IBNS as a homoplasy; a latent ability of all vertebrates with a central nervous system and the capacity for abstract cognition, where socio-ecological selective pressures for prosociality evoke the IBNS response. Future research into vertebrates outside the class Mammalia will provide further insight so assumptions made here are tentative. Generally, however, social adaptation to environmental cues appears to be the crux of IBNS. Therefore, by creating a framework where socio-ecological forces predicting prosocial behavior are a necessary condition to evoke IBNS, we offer a predictive model of IBNS which can be applied to all primates.
Past research in primates exploring the link between prosociality and environment mainly used cause-and-effect models of explanation (Tennie et al. 2016; Kuroshima and Fujita 2018; Shultz and Dunbar 2022). However, there is a lack of predictive correlational modelling and machine learning within this research area. This type of method would allow for a broader understanding of prosociality and IBNS in primates by creating probability estimates of prosocial behavior predicted by correlated socio-ecological variables. Predictive modelling and machine learning can use natural observations to calculate these probabilities and their stability when generalized from the model, which may broaden our knowledge of factors influencing the evocation of prosociality and IBNS in primates. In comparison, cause-and-effect models cannot predict behavioral relationships in the absence of an intervention and are restricted to explanatory analyses in niche or laboratory environments, detracting from generalizability (De Waal & Suchak 2010, Lopresti-Goodman & Villatoro-Sorto 2022). In behavioral research, establishing causality for a specific behavior requires a complete understanding of context and record of all environmental variables which is beyond the scope of the current study. Correlational models are therefore more effective at predicting outcomes as the statistical relationship between variables can be analysed and validated without a causal link (Allison 1999). Our exploratory study aims to achieve a broader conceptualization of IBNS in primates by using predictive modelling techniques to outline which generalizable prosocial behaviors could theoretically evoke IBNS (mediated by socio-ecological factors). Our findings may provide a foundation for both natural and experimental IBNS research in primatology, where so far, the topic has been neglected.
Method
We used data from existing literature to demonstrate the likely presence of IBNS in primates in multiple socio-ecological contexts. We first measured select prosocial behaviors. Forms of prosociality vary greatly and can become niche behaviors, reducing their generalizability between primate species and resulting in non-standardized measurements (Polit and Beck 2010). Accordingly, six prosocial behavioral traits that can be generally measured in primates were chosen: (i) consolation behavior, (ii) group-hunting, (iii) group-foraging, (iv) formation of coalitions, (v) food-sharing, and (vi) alloparenting. These prosocial variables are distinct behavioral markers of social cooperation and have been previously studied across primates (Boesch 2009; Burkart et al. 2014; De Waal and Van Roosmalen 1979; De Waal, Luttrell, and Canfield 1993; De Waal et al. 2008; De Waal and Suchak 2010; Hohn et al. 2024; Fuente et al. 2021; Van Leeuwen et al. 2021). We feel it is necessary to note here that we are not analysing specifically proactive prosociality, as all forms of prosocial behavior are currently theorized to evoke IBNS in H. sapiens. We correlated these six variables against forty-nine variables measuring social, ecological, morphological, and biological (SEMB) conditions.
The sample consisted of 41 primate species from a random stratified collection, representing approximately 8% of total extant primate species (NB: logit modelling only requires a sample of at least N = 20 for model stability; Concato et al. 1995). Stratification was necessary as information on individual primates is unbalanced; more popularly studied primates, such as the great apes or macaques, retain far more information on SEMB variables over less studied primates such as the Cacajao calvus or Galago senegalensis. Regardless, missing observations still occurred and were marked “N.A.” for easy translation into the R programming language. Power analyses are not necessary for this sample as there is no specific hypothesis that we are aiming to test, the data are non-explanatory, and we only aim to predict the occurrence of prosocial traits and IBNS based on associations between prosocial and SEMB variables. However, we offer hypotheses for further investigation based on the results of these predictions.
We gathered data on each primate species primarily from the Wisconsin National Primate Centre Primate Information Network (2023) and San Diego Zoo Wildlife Alliance Library (2023), two open-access databases which record primate information on behavior, biology, and ecology from observational studies. If no information on a specific variable existed in either of these databases, primary sources such as journal articles, academic books and textbooks were used to gather information on SEMB variables. These sources were accessed through keyword searches within the Zoological Record database. Objective, observational recordings of all primate data were dummy coded into arbitrary classifiers (ordinal or binary), while observations of variables on a continuous scale (e.g., amount of daily socialization) were not recoded as they need no categorical classification for statistical analysis. Some ordinal variables were dummy coded based on quartile distribution analyses. Predation pressure and resource competition variables were calculated from existing models which were used as predictive equations and were coded into the data table (van Schaik 1989; Hart 2007). A list of recodes can be found in Supplementary File 1 (Supplementary materials Table S1). A significant limitation of this dataset lies in the disparate observational sources used to inform it. There is no specific, standard reporting practice for observational studies of primates to follow, with varying quality and quantity of information between sources. This caveat introduces possible biases into the dataset, such as errors in reporting and lower reporting reliability based on more subjective interpretations of behavior.
All statistical analyses took place in R Studio (R Core Team 2024); all variables were imported from an initial CSV worksheet (Sevastidis, 2024). Naturally, the size of this dataset cannot be properly fitted into a binary logit model for each dependent variable (Harrell 2015). Thus, Kendall’s correlations were performed correlating each prosocial variable with all 52 independent SEMB variables. We used Kendall’s Tau (τ) for all correlations due to its flexibility in measuring non-normal variables of continuous, ordinal, and binary scales (Newson 2006). The lower τ-coefficient margin was set at 0.3 (fair association) to select associations between prosocial and SEMB variables that were suitable for predictive analyses using machine learning models. Variable selection through correlative analysis also assists in preventing the analysis of redundant variables where associations are multicollinear, which avoids over-fitting regression models (Montesinos-Lόpez, Montesinos-Lόpez and Crossa 2022). We set alpha (α) to 0.05 as the level of significance for all statistical analyses. Within binary predictive modelling of natural data, the confidence estimates of probability performance models are more relevant to identifying whether modelled values have occurred by random chance (Gardner and Altman 1986; Du Prel et al. 2009). Additionally, explicit testing of false positives using dedicated algorithms (e.g. AUROC) trumps the usage of an arbitrary p-value benchmark that is most applicable to two-tailed hypotheses, which are not investigated in this manuscript (merely offered for future research) (Kim and Bang, 2016; Jafari and Ansari-Pour 2018).
After we screened variable pairs via both correlation and significance measures, binary logit modelling using the prosocial variables could begin. Binary logit models were used for predictive modelling of binomial prosocial behavioral traits as dependent variables (Harrell 2015). We input each prosocial variable into its own logit regression with all SEMB traits that were significantly correlated to that prosocial variable. Generally, logit model equations cannot contain more than one predictor variable per ten observations in the dataset or they incur significant risk of overfitting the model and increasing Type 1 Error rates (Concato et al. 1995; Peduzzi et al. 1995). Since the dataset contains 41 total observations, it was only suitable to include four predictor variables in every model equation for each prosocial dependent variable. Thus, we used a combination of a priori knowledge of variable associations and step-AIC (Akaike Information Criterion) algorithm to remove excess predictor variables. We recommend that any step-wise method is used in conjunction with a priori analysis, as automatic functions can remove critical predictors from model equations (Heinze et al. 2015). Therefore, we formed models based on predictors previously associated with prosocial variables and comparisons between AIC values between-models, resulting in a best-fit model with the most relevant predictors. It should be noted here that removed predictors were significant, but less relevant than others. We checked models for multicollinearity through variance inflation factor (VIF) scores with an exclusion cut-off of 5; only variables below the exclusion cut-off were included in models (Marcoulides and Raykov 2019).
We used the initial model output to calculate the predicted probability of each prosocial behavior occurring in relation to its correlated socio-ecological (SEMB) variables. We obtained probabilities through exponentiation of modelled odds ratios. In models with more than one predictor variable, we used the full variation of all predictor value combinations to create predicted probabilities of prosocial behaviors associated with correlated socio-ecological (SEMB) variables. In models where two or more continuous predictor variables were present, separate model outputs were produced where each continuous predictor was variably held at its mean. This allowed deeper insight into each continuous variable’s association with a prosocial behavior by modelling its full range. We then plotted predicted probabilities of prosocial variables with associated SEMB variables using the ggplot2 package (Wickham 2016) in R Studio (R Core Team 2024). Where multiple variable values were missing or removed in a model predictor (due to statistical incompatibility), we used imputational binary logit modelling to establish estimates of likelihood and significance using the MICE package (Van Buuren and Groothuis-Oudshoorn 2011).
We reported McFadden’s pseudo-R2-values summarising the overall fit of each model using guidelines from Hensher and Stopher (1979), where McFadden reports ρ2-values between 0.2 and 0.4 a good fit (above 0.4 is considered an excellent fit). We also used confusion matrices to identify the ratio of false negatives and positives output by binary logit models, the results of which were displayed through fourfold plots for ease of interpretation.
Based on these values, we created an Area Under the Receiver Operating Characteristic Curve (AUROC) to display the overall accuracy of model predictions, comparing AUC scores with leave one out cross validation (LOOCV) computations for each model. Comparison to each model’s LOOCV AUC allows us to validate that predictive performance should remain stable when applied to similar populations external to the model. LOOCV uses N-1 iterations of the sample size for each model (40 iterations for each model) to train the model on all data points. The AUC output ranges within a 0-1 scale (compared against a random model at 0.5) and is a commonly used performance metric for model discrimination between specific classifiers. Confidence estimates of probability within the AUROC were distribution-independent as approximations on the link (logit) scale are unlikely to yield valid results for non-normal data, and binary data intervals have coverage equal to 0 (Cortes and Mohri 2005). Therefore, confidence estimates were avoided in binary logit models and were instead the focal point of AUROCs which reflect model accuracy of predicting true positives.
Results
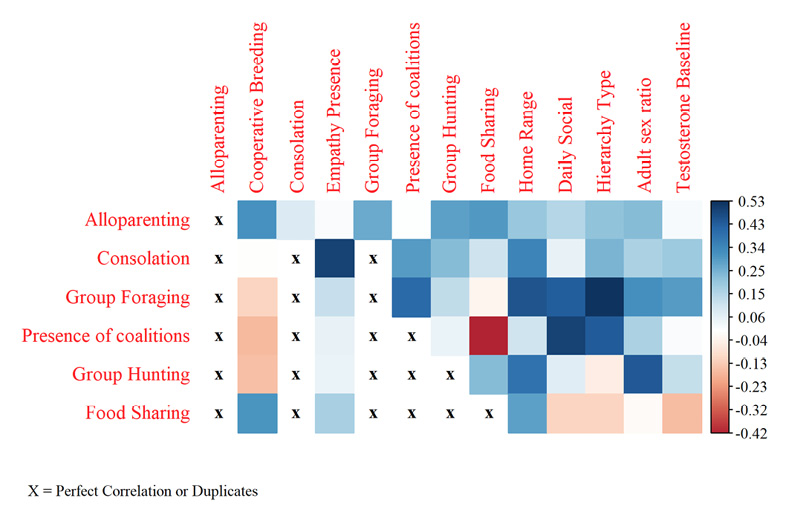
Correlation coefficients of selected SEMB variables to prosocial behaviors were all statistically significant (p = <0.05) (Sevastidis 2025). Due to the size of the correlation table, only variables included in modelling were displayed in the following heatmap (Figure 1).
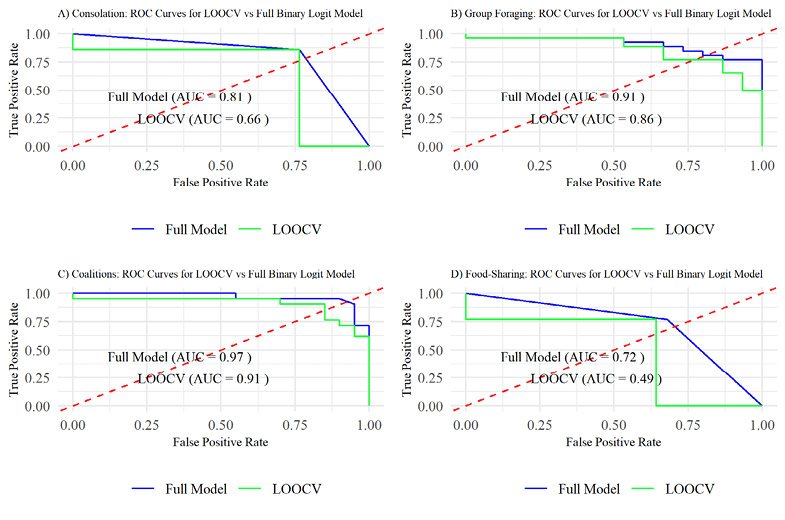
All results of original AUROC comparisons to LOOCV models can be seen below in Figure 2.
Alloparenting
Recorded observations of co-operative breeding in primates were not significantly predictive of alloparenting behavior (p = >0.05) (Supplementary materials Table S2). Perfect separation was detected between the variables, where all recorded co-operative breeding societies also displayed alloparenting behavior (Supplementary materials Figure S1). McFadden’s pseudo-R2 (ρ2 = 0.11), indicated poor model fit (Table B3).
Consolation
Consolation behavior in primate species was significantly predicted by observations of empathetic behavior; variables showed an excellent fit for the model (p = 0.01, ρ2 = 0.26) (Supplementary materials Tables S3 and S4). Observed empathetic behaviour was defined as any behaviors that are recorded in the literature as empathetic or observations of behaviors which are commonly described as empathetic (e.g. social contagions, targeted helping and so on) (Campbell and De Waal, 2014; Yamamoto, 2016). We did not include observations where empathy was assumed through abstract terms e.g. perspective taking, that did not accompany an accepted form of empathetic primate behavior. The absence of observed empathetic behavior resulted in a 3.7% probability that any given primate species would display consolation behaviors, while displays of empathy resulted in a 43% likelihood of consolation behaviors being present (Supplementary materials Table S5).
A four-fold plot displayed the rates of Type 1 and 2 Errors for the model (Supplementary materials Figure S2). Full model AUROC results indicated that the model has good predictive accuracy, outputting a true positive rate of 81% before producing any false positives (AUC = 0.81) (Figure 2A). However, once cross validated the AUC estimate was reduced to 0.66, indicating that while within-model prediction is excellent, applied to external populations predictions may only result in true positives 66% of the time. While confidence estimates for the full model are slim (95% CI = 0.8 to 1), the LOOCV model retains a wide interval (95% CI = 0.4 to 0.9) (Supplementary materials Table S6; generalizations from this model should be made cautiously.
Group Foraging
Both home range and daily socialising (as a % of a 24-hour day) significantly predicted the probability of group foraging behavior in primate species; model fit was excellent (home range p = 0.03, daily social p = 0.02, ρ2 = 0.48) (Supplementary materials Table S7). At their means, home range (M = 7.83km2) exceeded 7km2 and daily socialising (M = 5.84%) exceeded 5% of total time in the day, where primate species had a 93.4% probability of being group foragers. Since home range and daily socialising are continuous variables with a binary response, two separate plots were produced where one variable was held at its mean and vice versa (Supplementary materials Figure S3).
The AUROC for the full group foraging model inferred excellent predictive performance of the SEMB variables on group foraging behavior, outputting a true positive rate of 90.8% before encountering any Type 1 Errors (AUC = 0.908, 95% CI = 0.81 to 1) (Figure 2B, Supplementary materials Table S6). A Four-Fold plot of the binary confusion matrix showed that the model output four Type 1 and four Type 2 Errors out of 41 total predictions (Supplementary materials Figure S4). The LOOCV AUC comparison yielded a similar result (AUC = 0.86), validating the excellent predictive performance of this model when generalized to similar populations (95% CI = 0.74 to 0.96) (Supplementary materials Table S6).
Presence of Coalitions
Societies with no indication of strict despotism and egalitarianism were used as reference for these analyses. We aimed to measure predictive correlations of coalitionary behavior to distinct hierarchy types for clarity of analyses, but recognise that such behaviors are not universal to all primates. Daily socialising, food-sharing and despotic hierarchies were modelled with a statistically significant relationship to coalitionary behavior (daily social p = 0.04, food-sharing p = 0.04, hierarchy type 2 (despotic) p = 0.01, ρ2 = 0.51). Hierarchy type 3 (egalitarian) was not significantly related to coalitionary behavior in the model (p = 0.22), however is still important to be included in the model to classify data accurately (Supplementary materials Table S8). AUROCs displayed an excellent predictive accuracy for both full-model and the LOOCV model (full model AUC = 0.96, LOOCV AUC = 0.91) (Figure 2C) with high confidence estimates (full model 95% CI = 0.88 to 1, LOOCV 95% CI = 0.80 to 1) (Supplementary materials Table S6).
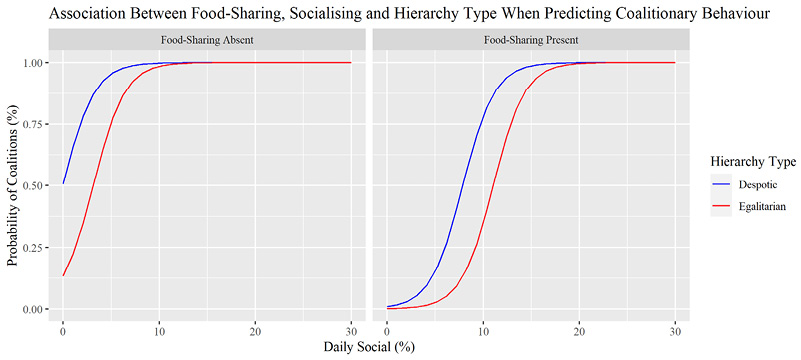
Figure 3 shows that food-sharing was a mediating variable influencing the ability of daily socialising to predict if primate species display coalitionary behavior. Food-sharing primates required higher levels of socialising to reach higher probabilities of coalition formation compared to primates where food-sharing has not been recorded. Additionally, rates of daily socialising reflecting similar predicted probabilities of coalition formation vary between hierarchy types and could indicate an inferential relationship to coalition formation in a despotic hierarchy. A Four-Fold confusion plot showed that the model produced two Type 1 and one Type 2 Errors (Supplementary materials Figure S5).
Group Hunting
Due to validity issues caused by missing values of testosterone baseline and adult sex ratio, imputational data were used to model both variables when predicting group hunting in primates. The imputed data resulted in both adult sex ratio and testosterone not being significant predictors of group hunting (adult sex ratio, p = 0.22; testosterone baseline, p = 0.60) shown in Table S9 (Supplementary materials). Density plots comparing iterations of imputed data showed that simulated values variably followed the general trend of real values within the original dataset (Supplementary materials Figure S6). A discussion on improving this model can be found in the Supplementary File 2.
Food Sharing
Food-sharing in primates was significantly predicted by the presence of coalitions (p = 0.01, ρ2 = 0.14) (Supplementary materials Table S10). Primates who display coalitionary behavior had a 13.6% probability of being food-sharing species, whereas the absence of coalitions resulted in a 52.6% probability of food-sharing in primates (Supplementary materials Table S11). AUROC results of the full model in Figure 2D displayed a good level of predictive accuracy (AUC = 0.72), however the LOOCV AUC displayed poor predictive accuracy for the model (AUC =0.49). Confidence estimates for the full model and LOOCV AUC are wide (95% CI = 0.57 to 0.87, LOOCV 95% CI = 0.28 to 0.7) (Supplementary materials Table S6), indicating a low reliability of predictions made both within and external to the model. A Four-Fold confusion plot showed that the model produced eleven Type 1 and three Type 2 Errors (Supplementary materials Figure S7).
Discussion
This research suggests that certain socio-ecological contexts may predict prosocial behaviors in primates. We proceed to discuss possible inferential relationships within our models and explore how prosociality within these contexts could provide a framework to test IBNS generally within this taxon. While the discussions on IBNS presented here are theoretical, it is hoped that our findings and hypotheses are tested through future IBNS research in primates, especially in species that naturally fulfil socio-ecological conditions that select for prosocial behaviors.
We note that alloparenting and co-operative breeding are erroneously used as interchangeable terms in much of the current primate literature. While the two are semantically similar, in this paper alloparenting is defined as a society with non-restrictive breeding and care provided by non-parents, whereas co-operative breeding is used for societies where alloparents are a by-product (or possibly forced consequence) of reproductive skew to dominant breeders (Lukas and Clutton-Brock 2012). However, the definitive usage for each term is still debated (Mocha et al. 2023).
The predictive relationship between co-operative breeding and alloparenting was not significant; considering the logical and frequently observed relationship between the two, this result seems surprising. However, the raw data suggest a perfect separation between the two variables. No co-operative breeding was recorded in primate societies without alloparenting; the statistical model could not calculate a significant difference between the two variables as no variance between them existed. Therefore, no predictive modelling could be extracted from these data, though readers should note that logically, a co-operative breeding society will always alloparent; otherwise, the purpose of co-operative breeding is lost and the behavior would likely not exist. Based on this assumption, we can infer that primate species which practice co-operative breeding also practice alloparenting. Future studies should consider exploring if distinct behaviors associated with alloparenting (which vary between primate species) have any correlations to SEMB variables and potentially IBNS (Cerrito and Spear 2022; Isler and van Schaik 2012).
Interplay Between Home Range and Daily Socialization When Predicting Group Foraging
Our study found that the likelihood of a primate being a group forager increased to nearly 100% when the daily time spent socialising was at 10% or more (Supplementary materials Figure S3a). Group or social foraging is a cohesive action that depends on physical and temporal co-ordination, making it an optimal variable to discriminate between primates that could hypothetically display IBNS. This also provides an opportunity for researchers to investigate if cortical synchronization is varied during IBNS in different co-operative tasks. While group foraging is a widespread and effective behavior in the animal world, its presence in primate species is variable (Swedell 2012). Group foraging behaviors can display uniquely in each primate and can depend on the decision of leading party members to secure vegetation (Strandburg-Peshkin et al. 2015; Williams et al. 2022).
Consider the group foraging methods of Pan paniscus. Mixed sex groups of five to six conspecifics forage for fruits in tall trees (Janson 1992); when fruits are secured, they can become euphoric and begin to chase, greet, copulate, vocalise, and eat (Kano and Mulavwa, 1984). Though not part of the actual foraging strategy, the process denotes a key part of group foraging behavior – social cohesion (Sterelny 2021; Williams et al. 2022). Consider another group foraging strategy from P. anubis; these primates travel and forage in large groups of up to 200 individuals (Kiffner et al. 2022). With such a large population, group foraging operates through party leaders, who decide the direction of the group and time spent at a single foraging site (Strandburg-Peshkin et al. 2015). Each group member must cooperate with the choices made by leaders of the foraging party to maximise individual foraging efficiency and reward in a species where home ranges can reach 19km2 (Barton et al. 1992). All group foraging is dependent on constant socialising and cooperation between group members. It is therefore unsurprising that group foraging forms a strong predictive relationship with daily time spent socialising (Supplementary materials Figure S3a). Group foraging behaviors in primates with high daily rates of socialization indicate an elevated social cohesiveness, thus making them optimal candidates to potentially observe IBNS between conspecifics. However, there are group foraging primates that reflect an inverse of the relationship displayed in (Supplementary materials) Figure S3a.
Lagothrix lagothricha and Erythrocebus patas are two examples of the conflicting patterns that emerge when using daily socialising to predict the presence of group foraging in primates. E. patas only spends 3% of its day socialising and is a group forager, whereas L. lagothricha spends 10% of its day socialising, but does not forage in groups. One explanation for this difference may be found in the home range of each primate; another predictive condition of group foraging behavior in primates. In Figure S3b (Supplementary materials), the model shows that when daily socialising is held at its mean, a wider home range predicts an increased probability of group foraging. L. lagothricha lives within a home range of approximately 0.6km2 compared to E. patas home range of approximately 4km2. In Figure S3a (Supplementary materials), a home range of 4km2 results in a group foraging probability estimate of 80%. Therefore, even when primates socialise for 10% or more of their day, their home range can potentially decrease the effect of daily socialising as a predictive condition for group foraging. On the other hand, primates who are not intense socialisers like E. patas are certainly not bereft of the ability to act prosocially. To use group foraging as an indicator of IBNS, ecological/behavioral predictive factors of home range and daily socialising must be considered together with the theoretical implications discussed here. These socio-ecological forces predicting group-foraging behaviors may also influence IBNS, potentially altering the number/loci of synchronized cortices between primates where a different motivator (i.e., the presence or absence of regular socialization) spurs IBNS. Specific analysis of the number of loci synchronized between conspecifics should be compared on a gradient from solitary to highly social primates; IBNS most likely exists on such a gradient underpinned by familiarity with conspecifics as seen in humans (Kinreich et al. 2017; Kurihara et al. 2024). Therefore, future research should also consider analysing IBNS within group-foraging primates like E. patas that are not highly social.
How Food-Sharing Could Mediate Coalition Formation in Different Primate Hierarchies
Figure 3 suggested that food-sharing may play a mediating role in the probability of primates displaying coalitionary behavior when measured with daily time spent socialising. Higher probabilities of coalition presence in non-food-sharing primates are achieved at lower levels of socialising compared to food-sharing primates. Hierarchical structure was also shown to be significant to this result. Generally, food-sharing in primates (which is rarely observed) serves one of two functions; to reinforce relationships (through reciprocity or ally consolidation) or to reduce harassment (tolerated theft) (Silk et al. 2013). Both require complex cognitive abilities including the capacity for reciprocity, understanding of social relationships and even anticipatory behavior of conspecifics, suggesting abstract thinking beyond immediate needs (Legg, Ostojić and Clayton 2014). To gain allies, elevated social tolerance between conspecifics (indexed by increased socialization) is required (Tai et al. 2012).
Top-down despotic societies create rigid social boundaries caused by dominance and kinship behaviors, which greatly narrow the opportunities for social relationships and limit socialising in general compared to egalitarian primates (Tombak et al. 2019). Typically, aggression reduces tolerance between conspecifics and forces subordinates in despotic societies to seek coping strategies, especially via social support in the form of coalitions (Sapolsky 2005). Considering this, a likely coalition where food-sharing would take place in despotic societies would be within kin and reciprocal relationships, as bachelor groups are competitive and prone to intra-aggression, and food-sharing is not known to take place during despotic reconciliation (Palagi and Norscia 2015). It must be noted here that food-type alone is not a predictor for any certain behavioral outcome (though it can be associated with certain behaviors) (Jaeggi and Schaik 2011).
Female-cryptic mate choice is an exemplary strategy that mixes both kinship and long-term investment in conspecifics, which can be mediated by food-sharing in both Pan troglodytes and M. mulatta (Dubuc et al. 2012; Crick et al. 2013). Firstly, by creating the illusion of paternity through sexual intercourse with variably ranked fathers, cryptic females can theoretically expand their social matrix and acquire multiple mate-guards. This inter-sexual coalition is highlighted by Sapolsky (2005) as a subordinate coping strategy in a despotic society. These relationships are usually prefaced by intense social affiliation such as grooming and physical contact (including sexual engagement), establishing a foundation for reciprocity following Bronsan and De Waal’s (2002) proposed three levels of reciprocity. In the highly despotic M. mulatta, female consorts are allowed to co-feed more often (Dubuc et al. 2012) at the same site as their male counterpart. In P. troglodytes, females receive higher quality food after copulation with a male food possessor even outside of peak fertility; this did not consolidate food-sharing with the same male for the next oestrus period (Crick et al. 2013). Therefore, by participating in bouts of intense socialization, despotic females can temporarily expand their social matrix and consolidate coalitions through transient reciprocal relationships which manipulate paternity, thus using food-sharing as a short-term investment strategy. We note here that the relationship between coalitionary behavior and egalitarian societies was not statistically significant. Accordingly, we discuss the non-relationship between egalitarianism and food-sharing predicting the presence of coalitions.
Primate egalitarianism strives to maintain higher levels of constant social tolerance between conspecifics (Boehm 1993; Pandit and Van Schaik 2003; Tombak et al. 2019; Harrod et al. 2020), thus eliminating the need for intense bouts of socialization to form coalitions. Therefore, should the need for coalitions ever arise (such as those seen in P. paniscus female coalitions against aggressive males) (Tokuyama and Furuichi 2016) preceding socialization (before coalitionary behavior) would theoretically be less intense compared to despotic primates as investment strategies that strengthen cohesion between conspecifics such as food-sharing could permanently exist as a feature of reciprocity in an egalitarian society (Jaeggi and Gurven 2013; Toombak et al. 2019). This makes egalitarian coalitionary support not a matter requiring increased bouts of intense socialising, but a social enforcement mechanism built on long-term investment in conspecifics which maintains egalitarianism. In H. sapiens, small-scale egalitarian societies such as the Hadza of Tanzania share food to maintain egalitarianism; with individuals levelling inter-group asymmetries in food income after acquisition (Pinheiro 2021). Therefore, while egalitarian long-term investment in conspecifics using food-sharing could contribute to coalitionary behavior, the basis of coalitions may change dependent on hierarchy type, so much so that a generalized inferential relationship between the two cannot be made. These differences between hierarchical contexts shaping coalition formation and food-sharing between primate populations reiterate that ultimately, prosocial behavior manifests in accordance with the overarching socio-ecological context. This reinforces the previously mentioned notion that IBNS in primates would also uniquely manifest based on the relationship between socio-ecological context and prosocial behavior; like H. sapiens (Sáez et al. 2015; Valencia and Froese 2020; Kurihara et al. 2024). We make this assumption in reference to our gradient theory of primate IBNS – social familiarity mediated by overarching socio-ecological factors will likely influence the manifestation and strength of IBNS in primates.
Why is Food-Sharing more Likely Outside of Primate Coalitions?
When predicting the probability of food-sharing using the presence of coalitions in primate societies, an inverse relationship was found indicating that food-sharing is a prosocial trait more likely to occur in the absence of any coalitionary behavior (Supplementary materials Table S11).
Though the model itself was unstable and cannot reliably predict the relationship between these two variables, it is worth noting that instances of food-sharing occur outside of primate coalitions e.g. tolerated theft and kin selection. The inferential relationship between food-sharing occurring outside of primate coalitions still has value to understanding different contexts underpinning prosocial primate behavior. However, it is apparent that food-sharing as a general label is not a suitable classifier to use in predictive analyses and we acknowledge this as a weakness of our model. Instead, we recommend that the food-sharing variable be further operationalized to reflect the distinct contexts in which this behavior takes place. Using a model with a more refined food-sharing variable may increase the accuracy and reliability of predicting the occurrence of this behavior in a general primate population.
Predicting Consolation Behaviors Through Empathy Presence in Primates
The presence of empathetic behavior in primates indicated a 40% likelihood of consolation behaviors being present within a primate species, whereas the absence of empathy inferred a less than 5% likelihood of any consolation behavior. It should be noted here that the empathy presence variable contained observations which reflected of both cognitive and affective empathy. Though either type of empathetic behavior can still lead to consolation, cognitive empathy has been shown in H. sapiens to motivate consolatory behavior to a greater extent (Abraham et al. 2014). Both cognitive and affective empathy have been theorized to underpin sympathetic concern and consolatory behaviors in great apes, but no data explicitly discriminating these factors exist (Clay and De Waal 2013; Webb et al. 2017). To evaluate the correlations/predictive relationships of empathy to consolation behaviors, more detailed data discriminating between cognitive and affective empathic styles in primates are needed. Furthermore, it is recommended that data on empathy are de-centralized from the great apes to achieve a wider perspective of empathic cognition in primates. Numerous studies in H. sapiens have detailed the connection between empathetic behavior and IBNS (Valencia and Froese 2020). Therefore, primates that display empathy and consolation behaviors could be optimal candidates to observe IBNS in future research.
Comparing Inter-Brain Neural Synchronization and Key Prosocial Behaviors Between Primates and Homo sapiens
So far, we have discussed predicted prosocial behaviors in primates which may guide researchers to species (outside of macaques) that exhibit unique patterns of IBNS in a range of socio-ecological contexts. We will go on to discuss the theoretical justification of IBNS related to these prosocial behaviors within primates, and identify key similarities and differences between H. sapiens and primate IBNS. We do this with the goal to highlight how IBNS may uniquely manifest with distinct prosocial behaviors influenced by environment and neurobiology in a range of primates.
Many of the prosocial variables presented in the discussion were predicted by daily socialising with an overarching theme of co-ordinated group activity. Group foraging and coalition formation are both behaviors that require large-scale co-operation to achieve common goals, underpinned by increased socialization. Similar patterns of prosociality are seen in modern H. sapiens, where group behaviors increase the success of common goals and efficiency of co-operation (Sinha et al. 2016; Szymanski et al. 2017). In H. sapiens, such group activities are underscored by IBNS elicited through socialization during co-operation (Valencia and Froese 2020). For example, coalitionary scenarios in H. sapiens (such as team sports) evoke IBNS via intense socialization (vocal/physical communication) and co-ordination to achieve a common goal of victory over opponents (an example of shared intentionality), potentially mediated by dopaminergic systems (Li et al. 2020; Liu et al. 2021; Probolovski and Dahan, 2021).
Considering the influence of cooperative group scenarios on the cortical manifestation of IBNS within H. sapiens, (Tseng et al. 2018; Valencia and Froese 2020) it is plausible that similar scenarios in non-human primates may elicit unique patterns of IBNS. When food-acquisition becomes the primary goal of primate co-operative teams (typically seen in group foraging primates) identical patterns of co-operation to H. sapiens may emerge. A primate group-foraging party may cooperate by synchronising behavior to commands from a party leader, with the common goal to increase foraging efficiency and success (a form of shared intentionality) (Hintz and Lonzarich 2018; Williams et al. 2022). Similar scenarios have been created with M. mulatta in a laboratory setting where observer-participant dyads completed motor courses to achieve food rewards (Tseng et al. 2018). Translation of these results to natural group foraging scenarios (where an even greater scale of teamwork is required in response to dynamic environments) would suggest that IBNS is active during group foraging, with activation patterns possibly being influenced by natural socio-ecological contexts.
However, distinct neuroendocrine systems differentially influence H. sapiens and primate prosocial behaviors which may evoke IBNS. Levels of dopamine (critical to H. sapiens prosociality) are much higher in the nucleus accumbens (NAcc) and striatum of H. sapiens compared to great apes (Raghanti et al. 2018; Hirter et al. 2021). Additionally, great apes retain a higher density of acetylcholine receptors (linked to dominance and territorialism) compared to H. sapiens (Raghanti et al. 2018). Interactive analyses have shown that dopamine in H. sapiens brain is modulated by both acetylcholine and glutamate and is responsible for increases in prosocial behavior (Lester et al. 2010; Sáez et al. 2015). Furthermore, increases in dopamine are rewarded through interaction with oxytocin, creating a distinct neurochemical positive feedback loop for prosociality in H. sapiens (Love 2013). Serotonergic systems in the orbito-frontal region of great apes also show correlation to pro-social behavior and inhibition of anti-social behaviors; comparatively hominins’ striatal serotonin levels show marked increases and correlations with prosocial behavior (Saniotis et al. 2021). The function and interaction of up-regulated oxytocin, dopaminergic and serotonergic systems within H. sapiens creates distinct enforcement mechanisms for co-operative and prosocial behavior, leading to distinct manifestations of IBNS (though these may not be critical to its elicitation; only cortical activation patterns) (Mu et al. 2016; Lotter et al. 2023).
In comparison, dopamine receptor modulation was shown to have no significant effect on prosociality in M. mulatta (Wendland et al. 2006). This suggests that IBNS could potentially be modulated by distinct neuroendocrine systems which selected for increased co-operation in H. sapiens (Clark and Henneberg 2021). Notably, cooperation is much less prominent in great apes compared to H. sapiens, highlighting a potential implication of differing neuroendocrine systems supporting prosociality (Tomasello 2023). However, the current literature on primate IBNS is extremely limited so far and no studies have presented direct evidence for a relationship between neurohormones and IBNS in primates. Group foraging practices of P. paniscus could provide future researchers with a case for dopamine-induced IBNS (in reference to implications of dopaminergic influence on primate IBNS patterns by Tsung et al. 2018) and vitally expand the existing literature on the neuroendocrine systems that could support primate IBNS.
However, prosocial activity does not always have to involve large coalitions or teams striving to achieve a common goal. Co-operation is also realized on a smaller scale in both the primates and H. sapiens in individuals who take on roles as alloparents or carers, sometimes subserved by empathic or socio-cultural motivations. Parenting in modern H. sapiens strongly elicits IBNS, mediated by communication and distress between parent and child (Liu et al. 2024), usually in the form of crying and distress vocalizations. Parent responses to infant distress are mediated by empathy in H. sapiens, particularly cognitive empathy (Abraham et al. 2014). Cortical areas responsible for cognitive empathy are synchronously activated in parental dyads by IBNS during observation of infant distress, an example of sympathetic concern (Wever et al. 2021). Sympathetic concern is followed by consolatory behavior in H. sapiens which evokes IBNS via inter-parental neural coupling (Liu et al. 2024). Consolation behavior motivated by IBNS and empathy has also been documented to increase social closeness in H. sapiens (Turner and Turner 2013).
Notably, H. sapiens parental concern-consolation reaction can result from several unique social scenarios involving infants and conspecifics; possibly following hominin behavioral adaptations due to erect posture and further development of the Default Mode Network (DMN). Early hominin erection of the trunk may have significantly reduced the tactile relationship between mother and infant, resulting in more frequent bouts of infant distress (via crying or vocalizations) richer in semantic meaning and empathic manipulation compared to great ape predecessors (Falk 2004). This may have led to the distinct behavioral displays of concern-consolation behaviors seen uniquely in H. sapiens such as feeding or motherese to alleviate distress, as well as the usage of distal communication to soothe infants (Falk, 2004; Hayashi and Matsuzawa 2017).
Primates also have the capacity for cognitive empathy and show sympathetic concern to socially close conspecifics in distress (De Waal 2008; De Waal 2011; Palagi et al. 2014), which can be followed by engagement in consolatory behaviors for the distressed (e.g., licking, grooming). Consolation behaviors in primates also predict improved social closeness and integration, reflecting the same pattern of empathy-motivated behavior seen in H. sapiens (Webb et al. 2017). However, consolation in this sense may occur through two different neuroanatomical pathways between primates and H. sapiens. The topography of H. sapiens and P. troglodytes prefrontal cortex shows distinct anatomical differences in relation to the DMN, namely the absence of the dorso-medial prefrontal cortex (D-MPFC) in P. troglodytes (Rilling 2014). The D-MPFC has functional relevance to language and theory of mind behaviors within H. sapiens, a distinct form of cognitive empathy which has been shown to facilitate IBNS (Decety 2010; Fishburn et al. 2018).
Although, great apes may lack this specific neuroanatomical pathway, they are still capable of using cognitive empathy that is rudimentary in comparison to H. sapiens (Hare et al. 2000). Advances in the anatomical development of the DMN may have occurred in early hominins with the development of language and more advanced forms of tool-making (Rilling 2014). This highlights the importance of Falk’s (2004) motherese hypothesis and Dissanayake’s (1999; 2003; 2004) work in hominin mother-infant interaction, when analysing the potential differences in patterns of cortical activity (during IBNS) between primates and H. sapiens. Notably, these works highlight an epoch in the hominin lineage where unique prosocial behaviors arose from exapted features of increasingly prosocial mother-infant interactions. This idea extends to neurohormonal regulation of prosociality (e.g., oxytocin pathways to regulate cohesion) and advancements in socio-cognitive architecture – including the DMN and advanced forms of empathy. Recent studies have compared the superior temporal sulcus (STS) of non-human primates to the temporoparietal junction (part of the DMN) in H. sapiens, particularly the circuit connecting the STS, amygdala and lateral intraparietal area in non-human primates which may be a precursor to the evolution of theory of mind in H. sapiens (Ong, Madlon-Kay and Platt 2021; Platt, Seyfarth and Cheney 2016). Focus should be given to brain areas showing protracted development correlated to prosocial behaviors as these are hypothesized to subserve increased prosocial interaction (and neural responsivity) in non-human primates and thus may be easier to measure for IBNS during prosocial interaction (Cerrito et al. 2024).
No studies to date have shown a relationship between theory of mind and IBNS in primates. It can be argued that neuroanatomical differences resulting from early hominin selection pressures have influenced the formation of distinct prosocial behaviors unavailable to other primates which elicit IBNS in H. sapiens. Considering that both primate and H. sapiens consolation behavior is activated via sympathetic empathy, especially towards infants, future studies should aim to investigate the presence of IBNS in alloparental primates who show high investment in the offspring of related individuals. It should be noted here that the current study did not use phylogenetic comparative analysis to correlate SEMB variables with complex prosocial behaviors, therefore limiting evolutionary tracing and analysis of them. However, we recommend that future research uses trait mapping and predictive modelling of paleoneurology associated with SEMB variables (see Pearson and Polly, 2023; Pearson and Polly 2024) to provide a richer description of behavioral phylogeny.
Using modelled predictions of prosociality in a diverse sample of primate species, we compared prosociality between primates and H. sapiens to explore the idea that IBNS may be possible in all primates. Primates behave within similar prosocial contexts that precede IBNS in H. sapiens, ranging from empathetic concern and consolation to group-based behaviors that require higher levels of socialising. Additionally, the current studies on primate IBNS clearly demonstrate the existence of this function within this taxon (though limited only to macaques), potentially underpinned by similar neurohormones to H. sapiens. However, key differences between these groups may exist in relation to how IBNS is evoked. The factors that differentiate IBNS between primates and H. sapiens have likely been shaped by early hominin selection pressures. This research has implications for investigating the presence of IBNS in hominins. In our study, we provide a framework on how IBNS could exist in all primate populations which fulfill socio-ecological conditions that elicit prosocial behaviors, and the potential distinctions of IBNS between primates and H. sapiens. We have provided multiple hypotheses for future researchers to test the existence of IBNS in a diverse range of primates; potentially expanding our understanding of IBNS in non-human primates, which could further extend our knowledge on IBNS in H. sapiens.
References
Abraham E, Hendler T, Shapira-Lichter I, Kanat-Maymon Y, Zagoory-Sharon O, Feldman R. 2014. Father’s brain is sensitive to childcare experiences. Proc Natl Acad Sci USA 111(27): 9792–9797. https://doi.org/10.1073/pnas.1402569111
Abraham E, Hendler T, Shapira-Lichter I, Kanat-Maymon Y, Zagoory-Sharon O, Feldman R. 2014. Father’s brain is sensitive to childcare experiences. Proc Natl Acad Sci USA 111(27): 9792–97. https://doi.org/10.1073/pnas.1402569111
Allison, P. D. 1999. Multiple regression: A primer. Pine Forge Press.
Amici F, Widdig A, MacIntosh AJ, Francés VB, Castellano-Navarro A, Caicoya AL, Karimullah K, Maulany RI, Ngakan PO, Hamzah AS, Majolo B. 2020. Dominance style only partially predicts differences in neophobia and social tolerance over food in four macaque species. Sci Rep 10(1): 22069. https://doi.org/10.1038/s41598-020-79246-6
Barton RA, Byrne RW, Whiten A. 1996. Ecology, Feeding Competition and Social Structure in Baboons. Behav Ecol Sociobiol 38(5): 321–29. http://www.jstor.org/stable/4601210
Beeler JA, Kisbye Dreyer J. 2019. Synchronicity: the role of midbrain dopamine in whole-brain coordination. eNeuro 6(2): ENEURO.0345–18.2019. https://doi.org/10.1523/ENEURO.0345-18.2019
Ben Mocha Y, Scemama de Gialluly S, Griesser M, Markman S. 2023. What is cooperative breeding in mammals and birds? Removing definitional barriers for comparative research. Biol Rev Camb Philos Soc 98(6): 1845–61.https://doi.org/10.1111/brv.12986
Boehm C. 1993. Egalitarian behavior and reverse dominance hierarchy. Curr Anthropol 34(3): 227–254. https://doi.org/10.1086/204166
Boesch C. 2009. The real chimpanzee: sex strategies in the forest. Cambridge University Press.
Boinski S, Garber PA .2000. On the move: how and why animals travel in groups. University of Chicago Press.
Boyd R, Richerson PJ. 2009. Culture and the evolution of human cooperation. Philos Trans R Soc Lond B Biol Sci 364(1533): 3281–88. https://doi.org/10.1098/rstb.2009.0134
Brattstrom BH. 1974. The evolution of reptilian social behavior. Am Zool 14(1): 35–49. https://doi.org/10.1093/icb/14.1.35
Brosnan SF, De Waal FB. 2002. A proximate perspective on reciprocal altruism. Hum Nat 13: 129–152. https://doi.org/10.1007/s12110-002-1017-2
Burkart JM, Allon O, Amici F, Fichtel C, Finkenwirth C, Heschl A, Huber J, Isler K, Kosonen ZK, Martins E, Meulman EJ, Richiger R, Rueth K, Spillmann B, Wiesendanger S, van Schaik SP. 2014. The evolutionary origin of human hyper-cooperation. Nat Commun 5(1), 4747. https://doi.org/10.1038/ncomms5747
Campbell MW, de Waal FB. 2014. Chimpanzees empathize with group mates and humans, but not with baboons or unfamiliar chimpanzees. Proc Biol Sci 281(1782), 20140013. https://doi.org/10.1098/rspb.2014.0013
Cerrito P, Gascon E, Roberts AC, Sawiak SJ, Burkart JM. 2024. Neurodevelopmental timing and socio-cognitive development in a prosocial cooperatively breeding primate (Callithrix jacchus). Sci Adv 10(44), eado3486. https://doi.org/10.1126/sciadv.ado3486
Cerrito P, Spear JK. 2022. A milk-sharing economy allows placental mammals to overcome their metabolic limits. Proc Natl Acad Sci USA 119(10), e2114674119. https://doi.org/10.1073/pnas.2114674119
Chaudhary N, Salali GD, Thompson J, Rey A, Gerbault P, Stevenson EG, Dyble M, E. Page A, Smith D, Mace R, Vinicius L. 2016. Competition for Cooperation: variability, benefits and heritability of relational wealth in hunter-gatherers. Sci Rep 6(1): 29120. https://doi.org/10.1038/srep29120
Clark G, Henneberg M. 2021. Cognitive and behavioral modernity in Homo erectus: Skull globularity and hominin brain evolution. Anthropol Rev 84(4): 467–85. https://doi.org/10.2478/anre-2021-0030
Clay Z, de Waal FB. 2013. Bonobos respond to distress in others: consolation across the age spectrum. PloS one 8(1): e55206. https://doi.org/10.1371/journal.pone.0055206
Concato J, Peduzzi P, Holford TR., Feinstein AR. 1995. Importance of events per independent variable in proportional hazards analysis I. Background, goals, and general strategy. J Clin Epidemiol 48(12): 1495–1501. https://doi.org/10.1016/0895-4356(95)00510-2
Cortes C, Mohri M. 2004. Confidence intervals for the area under the ROC curve. Adv Neural Inf Process Syst 17.
Crick J, Suchak M, Eppley TM, Campbell MW, De Waal FB. 2013. The roles of food quality and sex in chimpanzee sharing behavior (Pan troglodytes). Behaviour 150(11): 1203–24. https://doi.org/10.1163/1568539X-00003087
Darwin C. 1888. The descent of man: and selection in relation to sex. John Murray, Albemarle Street.
Dde Waal FB, Suchak M. 2010. Prosocial primates: selfish and unselfish motivations. Philos Trans R Soc Lond B Biol Sci 365(1553): 2711–22. https://doi.org/10.1098/rstb.2010.0119
De la Fuente MF, Sueur C, Garber PA, Bicca‐Marques JC, Souto A, Schiel N. 2022. Foraging networks and social tolerance in a cooperatively breeding primate (Callithrix jacchus). J Anim Ecol 91(1), 138–153. https://doi.org/10.1111/1365-2656.13609
De Waal F. 2011. Empathy in primates and other mammals. In: J Decety (ed.). Empathy. Cambridge, Massachusetts: MIT Press.
De Waal FB, van Roosmalen A. 1979. Reconciliation and consolation among chimpanzees. Behav Ecol Sociobiol 5: 55–66. https://doi.org/10.1007/BF00302695
De Waal FB, Leimgruber K, Greenberg AR. 2008. Giving is self-rewarding for monkeys. Proc Natl Acad Sci USA105(36): 13685–89.https://doi.org/10.1073/pnas.0807060105
De Waal FB. 1989. Food sharing and reciprocal obligations among chimpanzees. J Hum Evol 18(5): 433–59. https://doi.org/10.1016/0047-2484(89)90074-2
De Waal, FB, Luttrell, LM, Canfield, ME. 1993. Preliminary data on voluntary food sharing in brown capuchin monkeys. Am J Primatol, 29(1), 73–78. https://doi.org/10.1002/ajp.1350290108
Decety J. 2010. The neurodevelopment of empathy in humans. Dev Neurosci 32(4): 257–67. https://doi.org/10.1159/000317771
Dissanayake E. 1999. Antecedents of musical meaning in the mother-infant dyad. In: B Cooke and F Turner (eds.). Biopoetics: Evolutionary Explorations in the Arts. Lexington, Kentucky: ICUS. 367–97.
Dissanayake E. 2004. Motherese is but one part of a ritualized, multimodal, temporally organized, affiliative interaction. Behav Brain Sci 27(4): 512–13. https://doi.org/10.1017/S0140525X0432011X
Doody JS, Burghardt GM, Dinets V. 2013. Breaking the social–non‐social dichotomy: a role for reptiles in vertebrate social behavior research?. Ethology 119(2): 95–103. https://doi.org/10.1111/eth.12047
Dubois C, Call J, DeTroy S, Schütte S, Haun DB, Van Leeuwen EJ, Kaufhold SP. 2021. Chimpanzees behave prosocially in a group-specific manner. Sci Adv 7(9). https://doi.org/10.1126/sciadv.abc7982
du Prel JB, Hommel G, Röhrig B, Blettner M. 2009. Confidence interval or p-value?: part 4 of a series on evaluation of scientific publications. Dtsch Arztebl Int 106(19): 335–39. https://doi.org/10.3238/arztebl.2009.0335
Dubuc C, Hughes KD, Cascio J, Santos L R. 2012. Social tolerance in a despotic primate: Co‐feeding between consortship partners in rhesus macaques. Am J Phys Anthropol 148(1): 73–80. https://doi.org/10.1002/ajpa.22043
Falk D. 2004. Prelinguistic evolution in early hominins: Whence motherese? Behav Brain Sci 27(4): 491–503. https://doi.org/10.1017/S0140525X04000111
Fishburn FA, Murty VP, Hlutkowsky CO, MacGillivray CE, Bemis LM, Murphy ME, Huppert TJ, Perlman SB. 2018. Putting our heads together: interpersonal neural synchronization as a biological mechanism for shared intentionality. Soc Cogn Affect Neurosci 13(8): 841–49. https://doi.org/10.1093/scan/nsy060
Froese T, Loh CL, Putri F. 2024. Inter-brain desynchronization in social interaction: a consequence of subjective involvement?. Front Hum Neurosci 18: 1359841. https://doi.org/10.3389/fnhum.2024.1359841
Garber PA. 1987. Foraging Strategies among Living Primates. Annu Rev Anthropol 16: 339–64. http://www.jstor.org/stable/2155875
Gardner MJ, Altman DG. 1986. Confidence intervals rather than P values: estimation rather than hypothesis testing. Br Med J (Clin Res Ed), 292(6522): 746–50. https://doi.org/10.1136/bmj.292.6522.746
Gvirts-Probolovski HZ, Dahan A. 2021. The potential role of dopamine in mediating motor function and interpersonal synchrony. Biomedicines 9(4): 382. https://doi.org/10.3390/biomedicines9040382
Gvirts-Provolovski HZ, Perlmutter R. 2021. How can we prove the causality of interbrain synchronization? Front Hum Neurosci 15: 651949. https://doi.org/10.3389/fnhum.2021.651949
Haas R, Watson J, Buonasera T, Southon J, Chen JC, Noe S, Smith K, Viviano Llave C, Eerkens J, Parker G. 2020. Female hunters of the early Americas. Sci Adv 6(45): eabd0310. https://doi.org/10.1126/sciadv.abd0310
Hare B, Call J, Agnetta B, Tomasello M. 2000. Chimpanzees know what conspecifics do and do not see. Anim Behav 59(4): 771–85. https://doi.org/10.1006/anbe.1999.1377
Harrell FE. 2001. Volume 608: Regression modelling strategies: with applications to linear models, logistic regression, and survival analysis. 219–74. Springer.
Harrod EG, Coe CL, Niedenthal PM. 2020. Social structure predicts eye contact tolerance in nonhuman primates: evidence from a crowd-sourcing approach. Sci Rep 10(1): 6971. https://doi.org/10.1038/s41598-020-63884-x
Hart D. 2007. Predation on Primates: A Biogeographical Analysis. In: S Gursky-Doyen, & KAI Nekaris (eds.). Primate anti-predator strategies. New York, New York: Springer.
Hasson U, Frith CD. 2016. Mirroring and beyond: coupled dynamics as a generalized framework for modelling social interactions. Philos Trans R Soc Lond B Biol Sci 371(1693): 20150366. https://doi.org/10.1098/rstb.2015.0366
Hayashi M, Matsuzawa T. 2017. Mother–infant interactions in captive and wild chimpanzees. Infant Behav Dev 48: 20–29. https://doi.org/10.1016/j.infbeh.2016.11.008
Heinze G, Wallisch C, Dunkler D. 2018. Variable selection – A review and recommendations for the practicing statistician. Biometrical journal. Biom Z 60(3): 431–49. https://doi.org/10.1002/bimj.201700067
Henazi SP, Barrett L. 1999. The value of grooming to female primates. Primates 40: 47–59. https://link.springer.com/content/pdf/10.1007/BF02557701.pdf
Hensher DA, Stopher PR.1979. Behavioural Travel Modelling. 1st edition. Routledge. https://doi.org/10.4324/9781003156055
Hintz WD, Lonzarich DG. 2018. Maximizing foraging success: the roles of group size, predation risk, competition, and ontogeny. Ecosphere 9(10). https://doi.org/10.1002/ecs2.2456
Hirter KN, Miller EN, Stimpson CD, Phillips KA, Hopkins WD, Hof PR, Sherwood CC, Lovejoy CO, Raghanti MA. 2021. The nucleus accumbens and ventral pallidum exhibit greater dopaminergic innervation in humans compared to other primates. Brain Struct Funct 226(6): 1909–23. https://doi.org/10.1007/s00429-021-02300-0
Hoffmann S, Trost L, Voigt C, Leitner S, Lemazina A, Sagunsky H, Abels M, Kollmansperger S, Maat AT, Gahr M. 2019. Duets recorded in the wild reveal that interindividually coordinated motor control enables cooperative behavior. Nat Commun 10(1): 2577. https://doi.org/10.1038/s41467-019-10593-3
Hohn TI, Lin B, Miller CM, Foxfoot IR, Venkataraman VV, Ruckstuhl KE, Nguyen N, Fashing PJ. 2024. Post-Conflict Behaviors of Wild Gelada Monkeys (Theropithecus gelada) at Guassa, Ethiopia. Int J Primatol, 45(5), 1083–1106. https://doi.org/10.1007/s10764-024-00438-2
Hu Y, Hu Y, Li X, Pan Y, Cheng X. 2017. Brain-to-brain synchronization across two persons predicts mutual prosociality. Soc Cogn Affect Neurosci 12(12): 1835–44. https://doi.org/10.1093/scan/nsx118
Isler K, van Schaik CP. 2012. Allomaternal care, life history and brain size evolution in mammals. J Hum Evol, 63(1), 52–63. https://doi.org/10.1016/j.jhevol.2012.03.009
Jaeggi AV, Van Schaik CP. 2011. The evolution of food sharing in primates. Behav Ecol Sociobiol 65(11): 2125–40. https://doi.org/10.1007/s00265-011-1221-3
Jafari M, Ansari-Pour N. 2019. Why, when and how to adjust your p values? Cell J 20(4): 604–07. https://doi.org/10.22074/cellj.2019.5992
Janson CH. 1994. The last ape: pygmy chimpanzee behavior and ecology. Am Sci 82(4): 388–89.
Kano T, Mulavwa M. 1984. Feeding ecology of the pygmy chimpanzees (Pan paniscus) of Wamba. In: R Susman (ed.). The pygmy chimpanzee: Evolutionary biology and behavior. Boston, Massachusetts: Springer. 233–74.
Kiffner C, Paciência FM, Henrich G, Kaitila R, Chuma IS, Mbaryo P, Knauf S, Kioko J, Zinner D. 2022. Road-based line distance surveys overestimate densities of olive baboons. PloS one 17(2): e0263314. https://doi.org/10.1371/journal.pone.0263314
Kim J, Bang H. 2016. Three common misuses of P values. Dent Hypotheses 7(3): 73–80. https://doi.org/10.4103/2155-8213.190481
Kingsbury L, Huang S, Wang J, Gu K, Golshani P, Wu YE, Hong W. 2019. Correlated neural activity and encoding of behavior across brains of socially interacting animals. Cell 178(2): 429–46. https://doi.org/10.1016/j.cell.2019.05.022
Kinreich S, Djalovski A, Kraus L, Louzoun Y, Feldman R. 2017. Brain-to-Brain Synchrony during Naturalistic Social Interactions. Sci Rep 7(1): 17060. https://doi.org/10.1038/s41598-017-17339-5
Kopp KS, Liebal K. 2018. Conflict resolution in socially housed Sumatran orangutans (Pongo abelii). PeerJ 6: e5303. https://doi.org/10.7717/peerj.5303
Koski SE, Sterck EHM. 2010. Empathic chimpanzees: A proposal of the levels of emotional and cognitive processing in chimpanzee empathy. Eur J Dev Psychol 7(1): 38–66. https://doi.org/10.1080/17405620902986991
Kreider JJ, Kramer BH, Komdeur J, Pen I. 2022. The evolution of ageing in cooperative breeders. Evol Lett 6(6): 450–59. https://doi.org/10.1002/evl3.307
Kurihara Y, Takahashi T, Osu R. 2024. The topology of interpersonal neural network in weak social ties. Sci Rep 14(1): 4961. https://doi.org/10.1038/s41598-024-55495-7
Kuroshima H, Fujita K. 2018. Affective States, Motivation, and Prosocial Behaviour in Primates. In: LD Di Paolo, F Di Vincenzo, F De Petrillo (eds.). Evolution of Primate Social Cognition. Interdisciplinary Evolution Research, Vol. 5. Springer, Cham. https://doi.org/10.1007/978-3-319-93776-2_3
Legg EW, Ostojić L, Clayton NS. 2015. Food sharing and social cognition. Wiley Interdiscip Rev Cogn Sci 6(2): 119–129. https://doi.org/10.1002/wcs.1329
Lester DB, Rogers TD, Blaha CD. 2010. Acetylcholine-dopamine interactions in the pathophysiology and treatment of CNS disorders. CNS Neurosci Ther 16(3): 137–62. https://doi.org/10.1111/j.1755-5949.2010.00142.x
Li L, Wang H, Luo H, Zhang X, Zhang R, Li X. 2020. Interpersonal Neural Synchronization During Cooperative Behavior of Basketball Players: A fNIRS-Based Hyperscanning Study. Front Hum Neurosci 14: 169. https://doi.org/10.3389/fnhum.2020.00169
Liu T, Duan L, Dai R, Pelowski M, Zhu C. 2021. Team-work, team-brain: exploring synchrony and team interdependence in a nine-person drumming task via multiparticipant hyperscanning and inter-brain network topology with fNIRS. NeuroImage 237: 118147. https://doi.org/10.1016/j.neuroimage.2021.118147
Lopresti-Goodman SM, Villatoro-Sorto B. 2022. The benefits and challenges of conducting primate research in different settings. Animals 13(1), 133. https://doi.org/10.3390/ani13010133
Lotter LD, Kohl SH, Gerloff C, Bell L, Niephaus A, Kruppa JA, Dukart J, Schulte-Rüther M, Reindl V, Konrad K. 2023. Revealing the neurobiology underlying interpersonal neural synchronization with multimodal data fusion. Neurosci Biobehav Rev 146. https://doi.org/10.1016/j.neubiorev.2023.105042
Love TM. 2014. Oxytocin, motivation and the role of dopamine. Pharmacol Biochem Behav 119: 49–60. https://doi.org/10.1016/j.pbb.2013.06.011
Lu H, Wang X, Zhang Y, Huang P, Xing C, Zhang M, Zhu X. 2023. Increased interbrain synchronization and neural efficiency of the frontal cortex to enhance human coordinative behavior: A combined hyper-tES and fNIRS study. NeuroImage 282: 120385. https://doi.org/10.1016/j.neuroimage.2023.120385
Lukas D, Clutton-Brock T. 2012. Cooperative breeding and monogamy in mammalian societies. Proc R Soc Lond B Biol Sci 279(1736): 2151–56. https://doi.org/10.1098/rspb.2011.2468
Marcoulides KM, Raykov T. 2019. Evaluation of variance inflation factors in regression models using latent variable modeling methods. Educ Psychol Meas 79(5): 874–82. https://doi.org/10.1177/0013164418817803
Melis AP, Semmann D. 2010. How is human cooperation different? Philos Trans R Soc Lond B Biol Sci 365(1553): 2663–74. https://doi.org/10.1098/rstb.2010.0157
Miall DS, Dissanayake E. 2003. The poetics of babytalk. Human Nature 14: 337–64. https://doi.org/10.1007/s12110-003-1010-4
Mine JG, Slocombe KE, Willems EP, Gilby IC, Yu M, Thompson ME, Muller MN, Wrangham RW, Townsend SW, Machanda ZP. 2022. Vocal signals facilitate cooperative hunting in wild chimpanzees. Sci Adv 8(30). https://doi.org/10.1126/sciadv.abo5553
Montesinos López OA, Montesinos López A, Crossa J. 2022. Multivariate statistical machine learning methods for genomic prediction. Springer Nature. 71–108.
Mu Y, Guo C, Han S. 2016. Oxytocin enhances inter-brain synchrony during social coordination in male adults. Soc Cogn Affect Neurosci 11(12): 1882–93. 10.1093/scan/nsw106
Murray E, Wise S, Rhodes S. 2011. What Can Different Brains Do with Reward? In: Gottfried, J editor. Neurobiology of Sensation and Reward. Boca Raton, Florida: Taylor & Francis.
Newson R. 2006. Efficient calculation of jackknife confidence intervals for rank statistics. J Stat Softw 15: 1–10 https://doi.org/10.18637/jss.v015.i01
Ogawa Y, Shimada S. 2023. Inter-subject EEG synchronization during a cooperative motor task in a shared mixed-reality environment. Virt World 2(2): 129–143. https://doi.org/10.3390/virtualworlds2020008
Okada KI, Takeya R, Tanaka M. 2022. Neural signals regulating motor synchronization in the primate deep cerebellar nuclei. Nat Commun 13(1): 2504. https://doi.org/10.1038/s41467-022-30246-2
Ong WS, Madlon-Kay S, Platt ML. 2021. Neuronal correlates of strategic cooperation in monkeys. Nat Neurosci 24(1), 116–128. https://doi.org/10.1038/s41593-020-00746-9
Palagi E, Dall’Olio S, Demuru E, Stanyon R. 2014. Exploring the evolutionary foundations of empathy: consolation in monkeys. Evol Hum Behav 35(4): 341–49. https://doi.org/10.1016/j.evolhumbehav.2014.04.002
Palagi E, Norscia I. 2015. The season for peace: reconciliation in a despotic species (Lemur catta). PloS one 10(11): e0142150. https://doi.org/10.1371/journal.pone.0142150
Pandit SA, van Schaik CP. 2003. A model for leveling coalitions among primate males: toward a theory of egalitarianism. Behav Ecol Sociobiol 55: 161–68. https://doi.org/10.1007/s00265-003-0692-2
Pearson A, Polly PD. 2023. Temporal lobe evolution in extant and extinct Cercopithecoidea. J Mamm Evol 30(3), 683–694. https://doi.org/10.1007/s10914-023-09664-6
Pearson A, Polly PD. 2024. Temporal lobe evolution in Hominidae and the origin of human lobe proportions. Am J Biol Anthropol 185(4), e25027. https://doi.org/10.1002/ajpa.25027
Peduzzi P, Concato J, Feinstein AR, Holford TR. 1995. Importance of events per independent variable in proportional hazards regression analysis II. Accuracy and precision of regression estimates. J Clin Epidemiol 48(12): 1503–1510. https://doi.org/10.1016/0895-4356(95)00048-8
Pinheiro M. 2022. Egalitarian sharing explains food distributions in a small-scale society. J Artif Soc Soc Simul 25(3). https://doi.org/10.18564/jasss.4835
Platt ML, Seyfarth RM, Cheney DL. 2016. Adaptations for social cognition in the primate brain. Philos Trans R Soc Lond B Biol Sci 371(1687), 20150096. https://doi.org/10.1098/rstb.2015.0096
Polit DF, Beck CT. 2010. Generalization in quantitative and qualitative research: Myths and strategies. Int J Nurs Stud 47(11): 1451–58. https://doi.org/10.1016/j.ijnurstu.2010.06.004
Previc FH. 2009. The dopaminergic mind in human evolution and history. Cambridge: Cambridge University Press.
Primate Information Network (and publications therein).Wisconsin National Primate Centre. (n.d.). Retrieved 2023. https://primate.wisc.edu/primate-info-net/
R Core Team. 2021. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/.
Raghanti MA, Edler MK, Stephenson AR, Munger EL, Jacobs B, Hof PR, Sherwood CC, Holloway RL, Lovejoy CO. 2018. A neurochemical hypothesis for the origin of hominids. Proc Natl Acad Sci USA 115(6): E1108–E1116. https://doi.org/10.1073/pnas.1719666115
Ramakrishnan A, Ifft PJ, Pais-Vieira M, Byun YW, Zhuang KZ, Lebedev MA, Nicolelis MA. 2015. Computing arm movements with a monkey brainet. Sci Rep 5(1): 1–15. https://doi.org/10.1038/srep10767
Reimers L, Diekhof EK. 2015. Testosterone is associated with cooperation during intergroup competition by enhancing parochial altruism. Front Neurosci 9: 183. https://doi.org/10.3389/fnins.2015.00183
Rennung M, Göritz AS. 2016. Prosocial consequences of interpersonal synchrony. Z Psychol 224(3): 168–18. https://doi.org/10.1027/2151-2604/a000252
Rilling JK. 2014. Comparative primate neuroimaging: insights into human brain evolution. Trends Cogn Sci 18(1): 46–55. https://doi.org/10.1016/j.tics.2013.09.013
Rose MC, Styr B, Schmid TA, Elie JE, Yartsev MM. 2021. Cortical representation of group social communication in bats. Science, 374(6566): eaba9584. https://doi.org/10.1126/science.aba9584
Sáez I, Zhu L, Set E, Kayser A, Hsu M. 2015. Dopamine modulates egalitarian behavior in humans. Curr Biol 25(7): 912–19. https://doi.org/10.1016/j.cub.2015.01.071
Samuni L, Preis A, Mielke A, Deschner T, Wittig RM, Crockford C. 2018. Social bonds facilitate cooperative resource sharing in wild chimpanzees. Proc R Soc Lond B Biol Sci 285(1888): 1471–2954. https://doi.org/10.1098/rspb.2018.1643
San Diego Zoo Wildlife Alliance (and publications therein) (n.d.). Retrieved 2023. https://ielc.libguides.com/sdzg/main/journals_databases/databases
Saniotis A, Grantham JP, Kumaratilake J, Henneberg M, Mohammadi K. 2021. Going Beyond Brain Size. Anthropologie 59(1): 101–06. https://doi.org/10.26720/anthro.20.08.10.1
Sapolsky RM. 1990. Adrenocortical function, social rank, and personality among wild baboons. Biol Psychiatry 28(10): 862–878. https://doi.org/10.1016/0006-3223(90)90568-M
Sapolsky RM. 2005. The influence of social hierarchy on primate health. Science 308(5722): 648–52. https://doi.org/10.1126/science.1106477
Sevastidis J. 2024. Primate_Var_List_2025.xlsx (Version 3). The University of Adelaide. https://doi.org/10.25909/25895407.v3
Sevastidis J. 2025. Full List of Correlations for Predicting Prosociality in Primates: Socio-Ecological Influences and a Framework of Inter-Brain Neural Synchronization (Version 1). The University of Adelaide. https://doi.org/10.25909/28735580
Shultz S, Dunbar RI. 2022. Socioecological complexity in primate groups and its cognitive correlates. Phil Trans R Soc B 377(1860). https://doi.org/10.1098/rstb.2021.0296
Silk JB, Brosnan SF, Henrich J, Lambeth SP, Shapiro S. 2013. Chimpanzees share food for many reasons: the role of kinship, reciprocity, social bonds and harassment on food transfers. Anim Behav 85(5): 941–47. https://doi.org/10.1016/j.anbehav.2013.02.014
Sinha N, Maszczyk T, Wanxuan Z, Tan J, Dauwels J. 2016. EEG hyperscanning study of inter-brain synchrony during cooperative and competitive interaction. In 2016 Conf Proc IEEE Int Conf Syst Man Cybern (SMC) 4813–18. https://doi.org/10.1109/SMC.2016.7844990
Sterelny K. 2021. The Pleistocene social contract: Culture and cooperation in human evolution. Oxford University Press.
Strum SC.1981. Processes and products of change: baboon predatory behavior at Gilgil, Kenya. In: S Harding, & G Teleki (eds.). Omnivorous primates: Gathering and hunting in human evolution. New York Chichester, West Sussex: Columbia University Press. 255–302.
Swedell L. 2012. Primate sociality and social systems. Nature Education Knowledge 3(10).
Szymanski C, Pesquita A, Brennan AA, Perdikis D, Enns JT, Brick TR, Müller V, Lindenberger U. 2017. Teams on the same wavelength perform better: Inter-brain phase synchronization constitutes a neural substrate for social facilitation. NeuroImage 152: 425–36. https://doi.org/10.1016/j.neuroimage.2017.03.013
Tai YM, Chen MM, Zhang YH, Ma AX, Wang H, Wang X. 2022. Social Rank or Social Bonds: Which one Facilitates Coalition Formation in Male Tibetan Macaques? Biology 11(9): 1269. https://doi.org/10.3390/biology11091269
Tennie C, Jensen K, Call J. 2016. The nature of prosociality in chimpanzees. Nat Commun 7(1): 13915. https://doi.org/10.1038/ncomms13915
Titus AL, Knoll K, Sertich JJ, Yamamura D, Suarez CA, Glasspool IJ, Ginouves JE, Lukacic AK, Roberts EM. 2021. Geology and taphonomy of a unique tyrannosaurid bonebed from the upper Campanian Kaiparowits Formation of southern Utah: implications for tyrannosaurid gregariousness. PeerJ 9. https://doi.org/10.7717/peerj.11013
Tokuyama N, Furuichi T. 2016. Do friends help each other? Patterns of female coalition formation in wild bonobos at Wamba. Anim Behav 119: 27–35. https://doi.org/10.1016/j.anbehav.2016.06.021
Tomasello M. 2023. Differences in the Social Motivations and Emotions of Humans and Other Great Apes. Hum Nat 34(4): 588–604. https://doi.org/10.1007/s12110-023-09464-0
Tombak KJ, Wikberg EC, Rubenstein DI, Chapman CA. 2019. Reciprocity and rotating social advantage among females in egalitarian primate societies. Anim Behav 157: 189–200. https://doi.org/10.1016/j.anbehav.2019.09.010
Townsend C, Ferraro JV, Habecker H, Flinn MV. 2023. Human cooperation and evolutionary transitions in individuality. Philos Trans R Soc Lond B Biol Sci 378(1872). https://doi.org/10.1098/rstb.2021.0414
Townsend C, Ferraro JV, Habecker H, Flinn MV. 2023. Human cooperation and evolutionary transitions in individuality. Philos Trans R Soc Lond B Biol Sci 378(1872). https://doi.org/10.1098/rstb.2021.0414
Treves A. 2000. Theory and method in studies of vigilance and aggregation. Anim Behav 60(6): 711–722. https://doi.org/10.1006/anbe.2000.1528
Treves A. 2000. Theory and method in studies of vigilance and aggregation. Anim Behav 60(6): 711–722. https://doi.org/10.1006/anbe.2000.1528
Trumble BC, Smith EA, O’Connor KA, Kaplan HS, Gurven MD. 2013. Successful hunting increases testosterone and cortisol in a subsistence population. Proc R Soc B 281(1776): 20132876. https://doi.org/10.1098/rspb.2013.2876
Tseng PH, Rajangam S, Lehew G, Lebedev MA, Nicolelis MA. 2018. Interbrain cortical synchronization encodes multiple aspects of social interactions in monkey pairs. Sci Rep 8(1): 1–15. https://doi.org/10.1038/s41598-018-22679-x
Turner JB, Turner RJ. 2013. Social Relations, Social Integration, and Social Support. In: CS Aneshensel, JC Phelan, A Bierman (eds.). Handbook of the Sociology of Mental Health. Handbooks of Sociology and Social Research. Dordrecht, Netherlands: Springer.
Valencia AL, Froese T. 2020. What binds us? Inter-brain neural synchronization and its implications for theories of human consciousness. Neurosci Conscious 6(1). https://doi.org/10.1093/nc/niaa010
van Buuren S, Groothuis-Oudshoorn K. 2011. “mice: Multivariate Imputation by Chained Equations in R.” J Stat Softw 45(3): 1–67. https://doi.org/10.18637/JSS.V045.I03
van Houwelingen G, van Dijke M. 2023. Investing to gain others’ trust: Cognitive abstraction increases prosocial behavior and trust received from others. Plos one 18(4): e0284500. https://doi.org/10.1371/journal.pone.0284500
van Schaik CP. 1989. The Ecology of Social Relationships Amongst Female Primates. In: V Standen, & R Foley (eds.). Comparative Socioecology: the Behavioural Ecology of Humans and Other Mammals. Blackwell Scientific Publications.
Wang J, Meng F, Xu C, Zhang Y, Liang K, Han C, Gao Y, Yu X, Li Z, Zeng X, Ni J. 2025. Simultaneous intracranial recordings of interacting brains reveal neurocognitive dynamics of human cooperation. Nat Neurosci 28(1): 161–173. https://doi.org/10.1038/s41593-024-01824-y
Wass SV, Whitehorn M, Haresign IM, Phillips E, Leong V. 2020. Interpersonal neural entrainment during early social interaction. Trends Cogn Sci 24(4): 329–42. https://doi.org/10.1016/j.tics.2020.01.006
Webb CE, Romero T, Franks B, De Waal FB. 2017. Long-term consistency in chimpanzee consolation behaviour reflects empathetic personalities. Nat Commun 8(1): 292. https://doi.org/10.1038/s41467-017-00360-7
Wendland JR, Lesch KP, Newman TK, Timme A, Gachot-Neveu H, Thierry B, Suomi SJ. 2006. Differential functional variability of serotonin transporter and monoamine oxidase a genes in macaque species displaying contrasting levels of aggression-related behavior. Behav Genet 36: 163–172. https://doi.org/10.1007/s10519-005-9017-8
Wever MC, van Houtum LA, Janssen LH, Will GJ, Tollenaar MS, Elzinga BM. 2021. Neural signatures of parental empathic responses to imagined suffering of their adolescent child. NeuroImage, 232. 117886. https://doi.org/10.1016/j.neuroimage.2021.117886
Wickham H. 2016. ggplot2: Elegant Graphics for Data Analysis. Springer.
Williams L, Shultz S, Jensen K. 2022. The primate workplace: Cooperative decision-making in human and non-human primates. Front Ecol Evol 10. https://doi.org/10.3389/fevo.2022.887187
Wise SP. 2020. The evolution of the prefrontal cortex in early primates and anthropoids. In JH Kaas editor. Evolutionary neuroscience 2nd edition. Elsevier Academic Press. 669–707
Yamamoto S. 2017. Primate empathy: three factors and their combinations for empathy-related phenomena. Wiley Interdiscip Rev Cogn Sci 8(3), 10.1002/wcs.1431. https://doi.org/10.1002/wcs.1431
Zhang W, Rose MC, Yartsev MM. 2022. A unifying mechanism governing inter-brain neural relationship during social interactions. Elife 11 10.7554/eLife.70493
Zhang W, Yartsev MM. 2019. Correlated neural activity across the brains of socially interacting bats. Cell 178(2): 413–428. https://doi.org/10.1016/j.cell.2019.05.023
Zhu P, Liu W, Zhang X, Li M, Liu G, Yu Y, Li Z, Li X, Du J, Wang X, Grueter CC. 2023. Correlated evolution of social organization and lifespan in mammals. Nat Commun 14: 372. https://doi.org/10.1038/s41467-023-35869-7
Final information
Acknowledgements
We thank Dr. Scott Coussens for his contributions to discussions concerning the current research topic. This research was supported by the University of Adelaide, South Australia (Australia). We also thank the reviewers and editors of the International Journal of Primatology for their comments which helped revise the manuscript.
Conflict of interest
The authors declare that Maciej Henneberg is an Editor of Anthropological Review, but was not involved in the handling of this manuscript.
Author contributions
All authors contributed to study conceptualization, discussed results, and made critical contributions to revisions/editing, resulting in the final manuscript. JS was the lead researcher, conceived the study concept and design, acquired all data for data analysis, performed all data analysis, and wrote the entire manuscript. GC provided many materials for the study. MH contributed to the design of statistical methods.
Ethics statement
Not applicable.
Data availability
Data on primate species used for this study are freely accessible in online repositories (Wisconsin National Primate Research Center and San Diego Zoo Wildlife Alliance Library) or through online literature.
Funding
No funding was acquired for the current study.
Corresponding author
Jacob Sevastidis, School of Biomedicine, University of Adelaide, Adelaide, South Australia, 5005, Australia, e-mail: jacob.sevastidis@adelaide.edu.au
Supplementary materials
Supplementary File 1
| Variable Name | Meaning & Code |
|---|---|
| Inter/Intra Aggression | High = 1, Low = 2 |
| Physical or Threatening aggression | Threatening = 1, Physical = 2 |
| Affiliative behavior out-group | None = 1. Rare = 2, Common = 3 |
| Affiliative behavior within-group | Yes = 1, None = 2 |
| Pair bond | Absent = 1, Present = 2 |
| Coalition presence | Absent = 1, Present = 2 |
| Group hunting | Absent = 1, Present = 2 |
| Consolation | Absent =1, Present = 2 |
| Allo-parenting | Absent =1, Present = 2 |
| Food-sharing | Absent = 1, Present = 2 |
| Group-foraging | Absent = 1 Present = 2 |
| Infanticide Present | Absent = 1, Rare = 2, Common = 3, High = 4 |
| Predator number | Low = 1, Moderate = 2. High = 3 |
| Remain in natural habitat | None =1, Male = 2, Female = 3, Both = 4 |
| Diurnal or nocturnal | Nocturnal = 1, Diurnal = 2 |
| Location | Madagascar = 1, Africa = 2, Asia = 3, Neotropics = 4 |
| Habitat | Arboreal =1, Semi-terrestrial = 2, Terrestrial = 3 |
| Predation pressure | Very low = 1, Low = 2. Moderate =3, High = 4 (see Hart, D., 2007) |
| Solitary | No = 1, Yes = 2 |
| Resource Density | Low = 1, Seasonal = 2, High = 3 |
| Feeding comp | BGC = 1, WGS/BGC = 2, WGC/S = 3, WGC = 4, WGS/BGC/WGC= 5, WGS = 6 (see van Schaik, C. P. 1989). |
| Feeding Competition Intensity | None = 1, Low = 2, Seasonal = 3, High = 4 |
| Social Tolerance | Low =1, Mod =2, High =3 |
| Intrasexual Competition | None = 1, Low = 2, Common = 3, High = 4 |
| Primary Diet | Omnivorous =1, Folivorous = 2, Frugivorous = 3, Exudativore = 4, Graminivore = 5, Carnivore= 6 |
| Altricial/Precocial | Altricial = 1, Precocial = 2 |
| Sex. Dimorphism | None = 1, Low = 2, Moderate = 3, High = 4 |
| Co-operative Breeding | Absent =1, Present = 2 |
| Singular vs Plural Breeding | Singular = 1, Plural = 2 |
| Mating System | Polyandrous = 1, Monogamous, = 2, Polygynous = 3, Polygamy = 4 |
| Dominance Hierarchy | Fdominance = 1, M/Fdominance = 2, Mdominance = 3, None = 4 |
| Hierarchy type | Ambiguous =1, Despotic = 2, Egalitarian = 3 |
| Sexual segregation | Absent =1, Present = 2 |
| Empathy Presence | Absent = 1, Present = 2 |
| Recorded Empathy Type | Affective = 1, Cognitive = 2, Both = 3 |
| Tool use | Present (true use) =1, Present (proto use) = 2, Present (both) = 3 |
| brak danych | Estimate | Std. Error | Z-Value | Pr(>|Z|) |
|---|---|---|---|---|
| Intercept | 0.29 | 0.34 | 0.84 | 0.40 |
| Cooperative Breeding Presence | 18.28 | 2662.86 | 0.01 | 1.00 |
Pr(>|Z|) = P-value associated with Z-score.
Std.Error = standard error value.
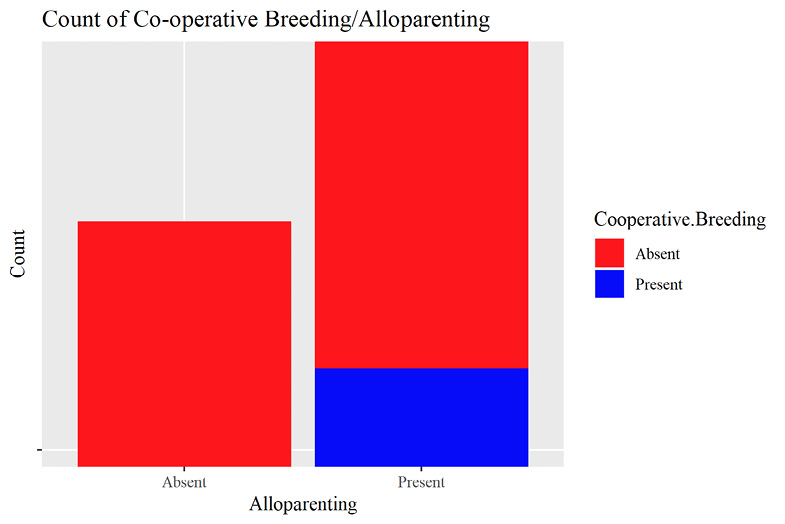
| brak danych | Consolation | Group Foraging | Presence of Coalitions | Group Hunting | Food Sharing | Allo-Parenting |
|---|---|---|---|---|---|---|
| McFadden’s ρ2 | 0.26 | 0.48 | 0.51 | 0.25 | 0.15 | 0.11 |
| brak danych | Estimate | Std. Error | Z-Value | Pr(>|Z|) |
|---|---|---|---|---|
| Intercept | -3.26 | 1.02 | -3.20 | 0.00 |
| Empathy Presence | 2.97 | 1.15 | 2.58 | 0.01 |
Pr(>|Z|) = P-value associated with Z-score.
Std.Error = standard error value.
| Empathy Presence | Fit | Se.fit | Residual Scale | UB | LB | Predicted Probability |
|---|---|---|---|---|---|---|
| Absent | -3.26 | 1.02 | 1.00 | 0.22 | 0.01 | 0.04 |
| Present | -0.29 | 0.54 | 1.00 | 0.68 | 0.21 | 0.43 |
Se.fit = Standard Error of Fit
UB = Upper boundary of 95% confidence interval
LB = Lower boundary of 95% confident
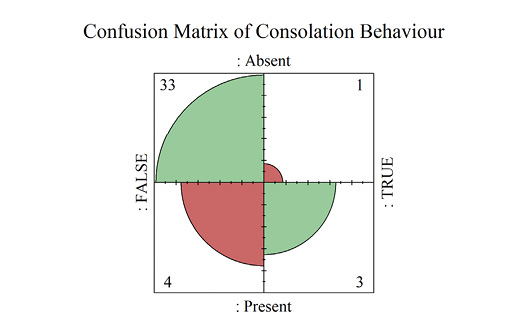
| AUC Consolation | LOOCV | AUC Food Sharing | LOOCV | AUC Group Foraging | LOOCV | AUC Presence of Coalitions | LOOCV | |
|---|---|---|---|---|---|---|---|---|
| LB | 0.80 | 0.41 | 0.58 | 0.29 | 0.81 | 0.75 | 0.89 | 0.80 |
| PE | 0.91 | 0.66 | 0.72 | 0.50 | 0.91 | 0.86 | 0.96 | 0.91 |
| UB | 1.00 | 0.90 | 0.87 | 0.70 | 1.00 | 0.98 | 1.00 | 1.00 |
LB = Lower boundary of 95% confidence interval
PE = Point estimate of 95% confidence interval
UB = Upper boundary of 95% confidence interval
LOOCV = Leave one out cross validation
| brak danych | Estimate | Std.Error | Z-value | Pr(>|Z|) |
|---|---|---|---|---|
| Intercept | -2.40 | 0.97 | -2.48 | 0.01 |
| Home Range | 0.37 | 0.17 | 4.14 | 0.03 |
| Daily Social | 0.37 | 0.16 | 2.33 | 0.02 |
Pr(>|Z|) = P-value associated with Z-score.
Std.Error = standard error value.
| brak danych | Estimate | Std.Error | Z-value | Pr(>|Z|) |
|---|---|---|---|---|
| Intercept | -4.10 | 1.68 | -2.44 | 0.02 |
| Daily Social | 0.60 | 0.29 | 2.05 | 0.04 |
| Food Sharing | -4.75 | 2.26 | -2.10 | 0.04 |
| Hierarchy(Despotic) | 4.13 | 1.60 | 2.58 | 0.01 |
| Hierarchy(Egalitarian) | 2.23 | 1.83 | 1.22 | 0.22 |
Pr(>|Z|) = P-value associated with Z-score.
Std.Error = standard error value.
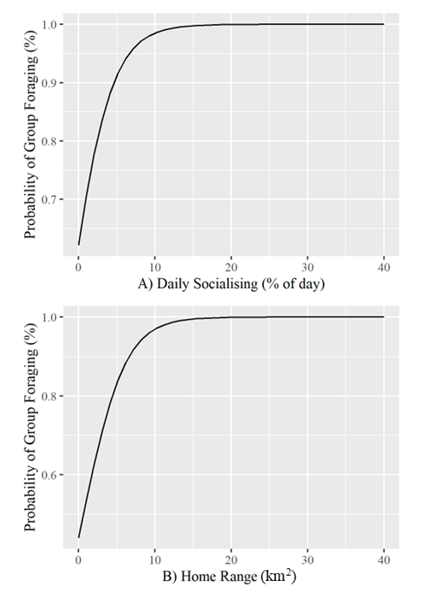
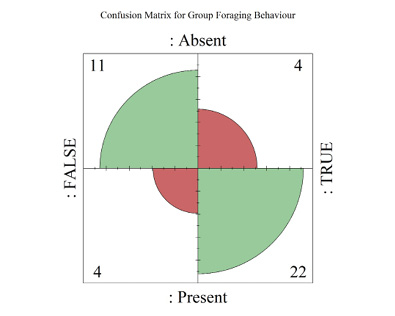
| Term | Estimate | Std.Error | Fit | Df | P.Value |
|---|---|---|---|---|---|
| Intercept | -2.86 | 1.33 | -2.15 | 30.74 | 0.04 |
| Adult Sex Ratio | 0.02 | 0.01 | 1.24 | 25.13 | 0.23 |
| Baseline Testosterone | -0.04 | 0.08 | -0.52 | 24.76 | 0.61 |
Df = Degrees of Freedom
Std.Error = Standard Error
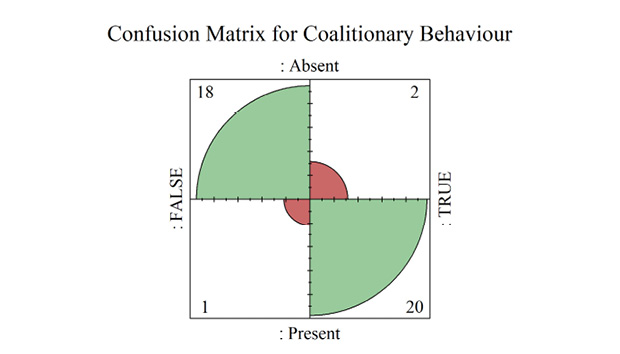
| Estimate | Std.Error | Z-value | Pr(>|Z|) | |
|---|---|---|---|---|
| Intercept | 0.11 | 0.46 | 0.23 | 0.82 |
| Presence of Coalitions | -1.95 | 0.77 | -2.53 | 0.01 |
Pr(>|Z|) = P-value associated with Z-score.
Std.Error = standard error value.
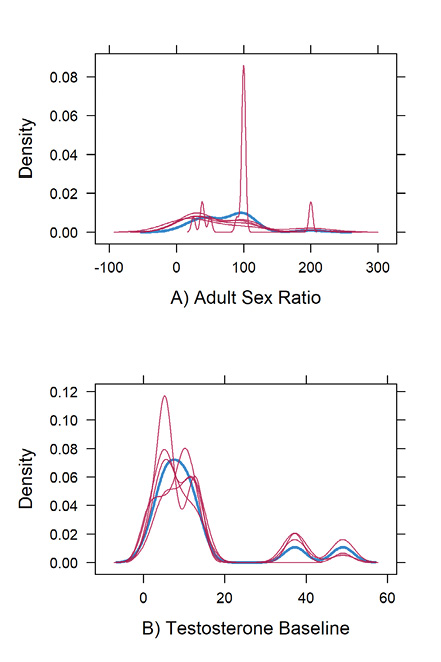
| Presence of Coalitions | Fit | Se.fit | Residual.Scale | UL | LL | Predicted Probability |
|---|---|---|---|---|---|---|
| Absent | 0.11 | 0.46 | 1.00 | 0.73 | 0.31 | 0.53 |
| Present | -1.85 | 0.62 | 1.00 | 0.35 | 0.05 | 0.14 |
Se.fit = Standard Error of Fit
UB = Upper boundary of 95% confidence interval
LB = Lower boundary of 95% confident
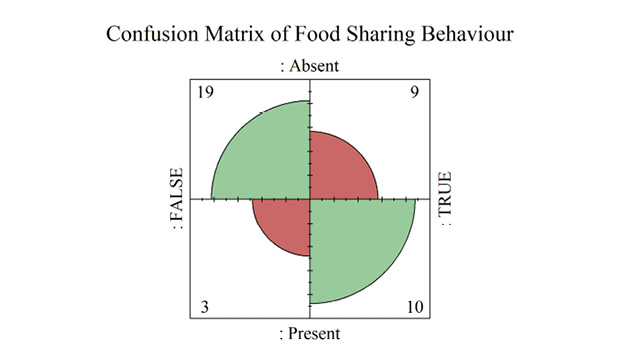
Supplementary File 2
Towards Better Predictors for IBNS and Group Hunting in Primates
There exist limited data on testosterone levels and adult sex ratios in many primates within our sample; limiting statistical analyses. Therefore, imputation was used to continue the analysis with these variables. While imputation is commonly used in machine learning analyses, it carries a significant risk of distorting ordinal data (Alam 2023). Adult sex ratio and testosterone were not significant predictors of group hunting in primates based on simulated data (Table S10). Reliability tests showed varied adherence to original data and so the pooled results of imputed logit models should be taken with caution (Figure S6a and S6b). Currently, we did not find any empirical evidence that supported a relationship between higher levels of testosterone and group-hunting. Group-hunting is a primarily co-operative behavior that likely uses neuroendocrine (dopamine) systems associated with reward and co-ordination (Samuni et al. 2018). However, this does not imply that testosterone is unrelated to hunting behavior (Trumble et al. 2014), nor that it is irrelevant to other displays of in-group co-operation (Reimers and Diekhof 2015). Ultimately, testosterone is an unlikely factor that could predict group-hunting. Dopamine presents a possible avenue for predicting group hunting in primates and would potentially show more association with IBNS (Previc 2009).
Group-hunting in primates is not strictly unisexual (Strum 1981; Gilby et al. 2015; Klein et al. 2021). This trend is also reflected in hominins, with recent research challenging the man-the-hunter hypothesis (Haas et al. 2020). However, this should not rule out adult sex ratio as a predictive variable of group-hunting in primates. Considering the effects of adult sex ratio on male-male mating competition (Darwin 1871) along with the fact that hunting ability influences mating access in P. troglodytes and hominins (Crick et al. 2013; Chaudhary et al. 2016), male adult sex ratio could be a plausible factor in predicting group-hunting in primates. However, looking at adult sex ratio and group hunting from the male perspective is one-dimensional and could limit predictions of group hunting in primates. In P. troglodytes, group-hunting probability increases with more females present; however, this statistic may be influenced by seasonal breeding and age-rank (Gilby et al. 2015). Therefore, it is likely a combination of mating competition, rank order, and adult sex ratio of both sexes which could potentially predict the likelihood of group hunting in primates. The dopaminergic system in apes facilitates communication and cooperation, underpinning motivation for exploration and problem-solving; enabling adaptation to a dynamic environment which is key in group-hunting efforts such as those seen in P. troglodytes (Previc 2009; Mine et al. 2022). Analysis of group-hunting probability using the SEMB variables listed above should be carried out in future research. Predictions of group-hunting using this combination of variables will likely reveal which primates should be investigated to observe IBNS evoked by group-hunting. Ultimately, this may reveal unique topographic IBNS patterns specific to cortical areas associated with group-hunting in primates.
References
Alam S, Ayub MS, Arora S, Khan MA. 2023. An investigation of the imputation techniques for missing values in ordinal data enhancing clustering and classification analysis validity. Decis Anal J 9:100341. https://doi.org/10.1016/j.dajour.2023.100341
Chaudhary N, Salali GD, Thompson J, Rey A, Gerbault P, Stevenson EG, Dyble M, E. Page A, Smith D, Mace R, Vinicius L. 2016. Competition for Cooperation: variability, benefits and heritability of relational wealth in hunter-gatherers. Sci Rep 6(1): 29120. https://doi.org/10.1038/srep29120
Darwin C. 1888. The descent of man: and selection in relation to sex. John Murray, Albemarle Street.
Gilby IC, Machanda ZP, Mjungu DC, Rosen J, Muller MN, Pusey AE, Wrangham RW. 2015. ‘Impact hunters’ catalyse cooperative hunting in two wild chimpanzee communities. Proc R Soc Lond B Biol Sci 370(1683): 20150005. https://doi.org/10.1098/rstb.2015.0005
Haas R, Watson J, Buonasera T, Southon J, Chen JC, Noe S, Smith K, Viviano Llave C, Eerkens J, Parker G. 2020. Female hunters of the early Americas. Sci Adv 6(45): eabd0310. https://doi.org/10.1126/sciadv.abd0310
Klein H, Bocksberger G, Baas P, Bunel S, Théleste E, Pika S, Deschner T. 2021. Hunting of mammals by central chimpanzees (Pan troglodytes troglodytes) in the Loango National Park, Gabon. Primates 62(2): 267–78. https://doi.org/10.1007/s10329-020-00885-4
Mine JG, Slocombe KE, Willems EP, Gilby IC, Yu M, Thompson ME, Muller MN, Wrangham RW, Townsend SW, Machanda ZP. 2022. Vocal signals facilitate cooperative hunting in wild chimpanzees. Sci Adv 8(30). https://doi.org/10.1126/sciadv.abo5553
Previc FH. 2009. The dopaminergic mind in human evolution and history. Cambridge: Cambridge University Press.
Reimers L, Diekhof EK. 2015. Testosterone is associated with cooperation during intergroup competition by enhancing parochial altruism. Front Neurosci 9: 183. https://doi.org/10.3389/fnins.2015.00183
Samuni L, Preis A, Mielke A, Deschner T, Wittig RM, Crockford C. 2018. Social bonds facilitate cooperative resource sharing in wild chimpanzees. Proc R Soc Lond B Biol Sci 285(1888): 1471–2954. https://doi.org/10.1098/rspb.2018.1643
Strum SC.1981. Processes and products of change: baboon predatory behavior at Gilgil, Kenya. In: Harding, S., & Teleki, G, editors. Omnivorous primates: Gathering and hunting in human evolution. New York Chichester, West Sussex: Columbia University Press. 255–302
Trumble BC, Smith EA, O’Connor KA, Kaplan HS, Gurven MD. 2013. Successful hunting increases testosterone and cortisol in a subsistence population. Proc R Soc B 281(1776): 20132876. https://doi.org/10.1098/rspb.2013.2876